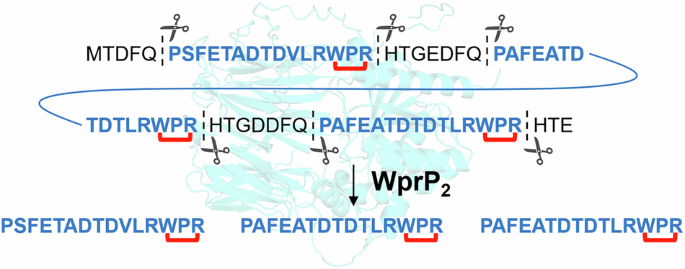
Scientists studying ribosomally synthesized and post-translationally modified peptides, or RiPPs, have uncovered a new serine protease that broadens what is known about precursor peptide processing. The enzyme, called WprP2, comes from Streptomyces venezuelae NPDC049867 and sits next to the radical SAM enzyme WprB2 in a gene cluster linked to cyclophane natural product biosynthesis.
In a new characterization study, WprP2 was shown to cleave the precursor peptide WprA2 in a highly uniform manner. That behavior stands out because it does not match the cleavage patterns typically seen for serine proteases involved in RiPP maturation.
Why this matters
RiPP biosynthesis depends on proteases that remove leader regions from precursor peptides so the mature natural product can form. While cysteine proteases, metalloproteases, and several serine protease families are already known in this space, the S9 family has been relatively small. Before this report, only a few S9 proteases had been linked to RiPP pathways.
WprP2 expands that family and suggests that S9 proteases may play a wider role in RiPP assembly than previously appreciated. Its cleavage mode also appears distinct from the better-known S8 proteases and from other S9 enzymes reported so far.
Link to cyclophane biosynthesis
The discovery is especially interesting because it ties proteolysis to a radical SAM-dependent pathway. Radical SAM enzymes are increasingly recognized for building unusual peptide cross-links, including cyclophane-like structures. In many of these pathways, the relevant protease has not been obvious, which has made it harder to fully reconstruct how the final natural products are made.
By pairing WprP2 with WprB2 in the same biosynthetic neighborhood, the study provides a more complete picture of how this cyclophane-related RiPP may be assembled. It also highlights how much peptide-processing chemistry remains to be discovered in bacterial natural product gene clusters.
Takeaway
WprP2 is more than another protease annotation. It is a newly characterized S9 serine protease with an unusual, uniform cleavage activity on a RiPP precursor peptide. That makes it a useful addition to the RiPP toolkit and a reminder that peptide biosynthesis still holds many surprises.