A newly characterized protease is expanding what scientists know about peptide maturation in ribosomally synthesized and post-translationally modified peptides, or RiPPs.
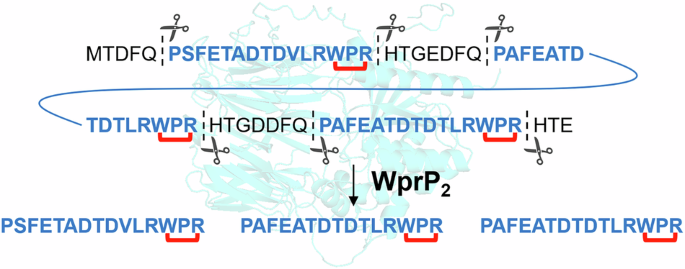
In work published in Communications Chemistry, researchers report that WprP2, an uncharacterized S9 serine protease from Streptomyces venezuelae NPDC049867, sits near the radical SAM enzyme WprB2 in a gene cluster linked to cyclophane natural product biosynthesis. When the enzyme was tested in vitro, it cleaved the precursor peptide WprA2 in a highly uniform manner.
That detail matters because proteases in RiPP pathways usually show defined but limited cleavage behaviors. The study suggests WprP2 represents a cleavage mode not previously observed among serine proteases involved in RiPP assembly, adding a new functional example to the small S9 family.
RiPPs rely on a two-step logic: enzymes first install chemical modifications on a peptide precursor, then a protease removes the leader region to release the mature product. In many pathways, that processing step is essential for biological activity. The new findings broaden the roster of proteases known to participate in this final maturation stage.
More broadly, the result may help researchers decode other orphan RiPP gene clusters, especially those involving radical SAM enzymes where the matching processing protease is not obvious from sequence alone.
As the field maps more of these unusual biosynthetic systems, enzymes like WprP2 could become useful clues for predicting how complex peptide natural products are assembled.