Elastin-like polypeptides are best known for their unusual response to environmental cues such as heat and salt. Built from repeating VPGXG motifs, these biomaterials can shift between more soluble and more compact states, making them attractive for sensing, delivery, and other engineered applications.
But one key question has remained difficult to answer: can the transition behavior of very short elastin-like peptides still be detected once they are fixed to a surface? A new study tackles that challenge with an electrochemical readout designed specifically for surface-bound peptides.
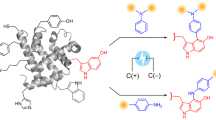
The researchers attached short engineered elastin-like sequences to gold electrodes through an N-terminal cysteine tag. They then added a C-terminal tyrosine tag, giving each peptide a built-in electroactive handle. Because tyrosine can be oxidized at an appropriate voltage, the team used cyclic voltammetry to monitor whether changes in peptide conformation altered the measured current.
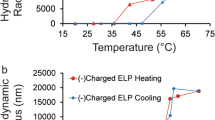
As the short peptides responded to changing conditions, their electrochemical signal shifted in a way that reflected the underlying transition. The effect depended on peptide design, including differences in hydrophobicity, suggesting that sequence composition still plays a major role even in these compact surface-tethered constructs.
To support the electrochemical findings, the team also examined the same peptides in solution using UV-visible spectroscopy. Comparing the two readouts helped distinguish behavior that was intrinsic to the peptide sequence from effects introduced by surface attachment.
The broader significance of the work is methodological: it shows that a simple oxidation-based signal can be used to study conformational switching in short elastin-like peptides on gold. That could make it easier to characterize thermoresponsive peptide systems in biosensing, surface engineering, and other applications where immobilization is unavoidable.
More generally, the study adds to growing interest in using peptide-based materials as programmable, stimulus-responsive building blocks. By pairing a classic electrochemical technique with a cleverly tagged peptide design, the authors provide a practical route for probing transitions that have been difficult to observe directly on surfaces.