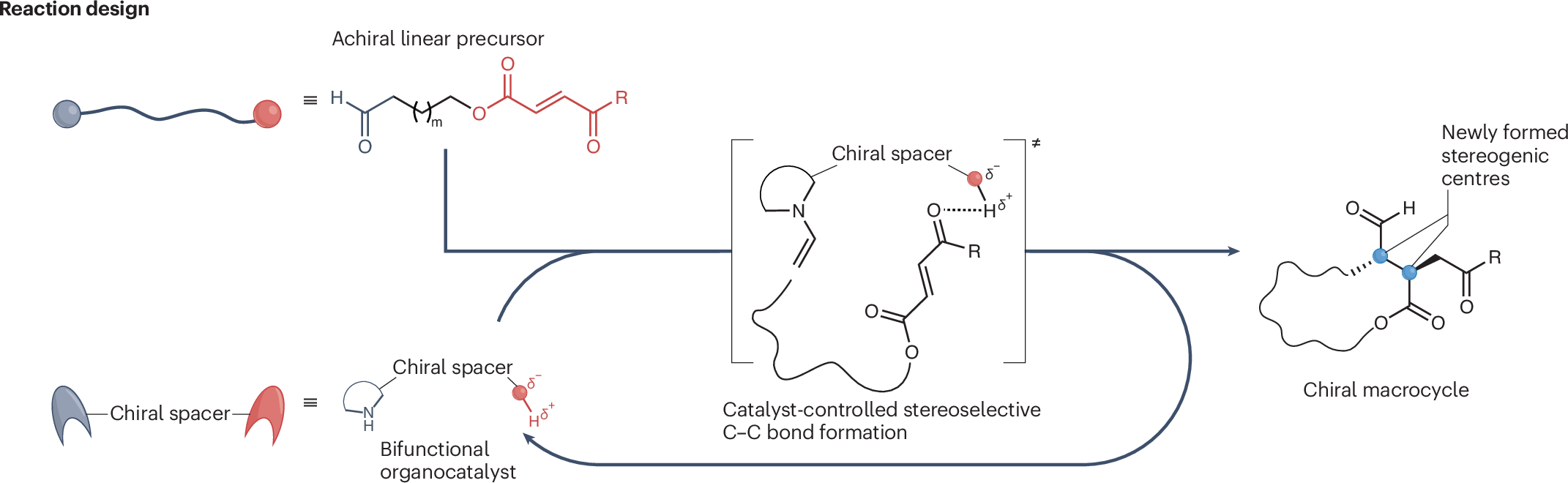
A new study highlights a peptide synthesis approach designed to make one of chemistry’s most widely used methods more water-friendly. Donald A. Wellings, Morten Meldal, John D. Wade and co-workers report a solid-phase peptide synthesis (SPPS) workflow built around a hydrophilic, biodegradable poly-ε-lysine-based support and aqueous reaction conditions.
The key enabling step is the conversion of conventional Nα-Fmoc amino acids into water-soluble salts before coupling. The team found that pre-treatment with bases such as N-methylmorpholine (NMM) or N,N,N-triethanolamine (TEOA) allows high-concentration aqueous formulations, extending the strategy across 20 standard Fmoc-protected amino acids.
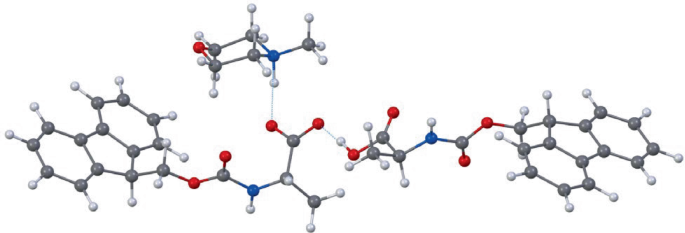
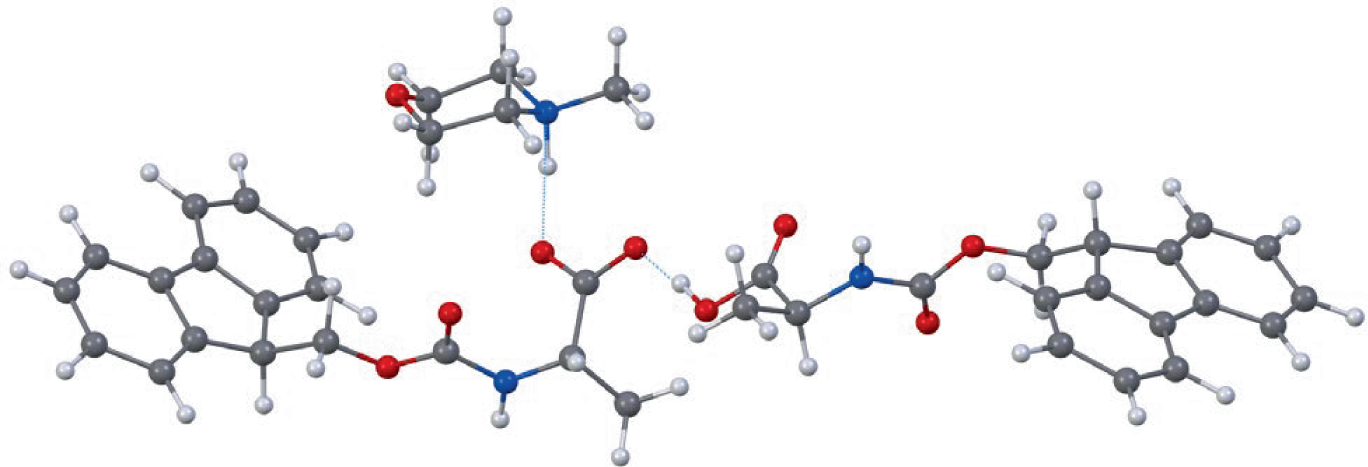
To better understand the chemistry behind the solubility boost, the researchers used 3D electron diffraction on the resulting amino acid–base crystals. The structural data supported a 1:1 relationship between the amino acid and base, with evidence that the amine component is deprotonated within the complex.
While classic SPPS still depends heavily on solvent-intensive workflows, this work points toward a more sustainable alternative by pairing water-compatible building blocks with a biodegradable support. If broadly adaptable, the method could help lower solvent use without sacrificing the familiar Fmoc chemistry used in peptide assembly.
For peptide researchers, the most interesting part may be that the chemistry does not require a complete reinvention of synthesis logic. Instead, it reframes familiar reagents in a water-based system, suggesting a practical path toward greener peptide manufacturing.