Getting DNA into the nucleus is one of the biggest bottlenecks in non-viral gene delivery. That challenge becomes even more pronounced in growth-arrested cells, where the usual route of nuclear entry during cell division is largely unavailable. In a new study, researchers describe a modular peptide-conjugation workflow that helps DNA cargo cross this barrier more effectively.
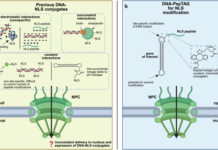
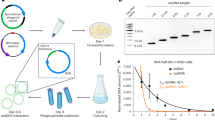
The team built peptide-modified gene cassettes by first generating DNA oligonucleotide-peptide conjugates with an enzymatic tagging system based on E. coli tRNA guanine transglycosylase, then ligating those units onto linear DNA. The result was a DNA-peptide platform, referred to as DNA-PepTAG, designed to present nuclear localization signal (NLS) peptides in a more controlled and reproducible way.
When the researchers delivered an eGFP reporter by lipofection into growth-arrested cells, NLS-modified cassettes showed stronger nuclear localization, higher mRNA output, and greater protein expression than unmodified DNA. In the best cases, expression increased by roughly an order of magnitude.
One important finding was that not all NLS peptides performed equally across cell types. Screening in several human growth-arrested cell lines revealed clear cell-type preferences for DNA nuclear transport. Still, two candidates stood out: PLSCR-1 and extSV40 produced consistently strong expression across the tested systems, suggesting they may be particularly useful for broader applications.
The approach was also tested with different payloads, including genes encoding both cytosolic and secreted proteins, as well as gene cassettes ranging from about 1.3 kbp to 7 kbp. This range suggests the method is not limited to a single reporter construct and may be adaptable to a variety of therapeutic or research DNA cargos.
For peptide researchers, the study is a useful reminder that peptide identity is only part of the story. How a peptide is attached to DNA can strongly influence whether it can do its job. By offering a site-specific, modular conjugation strategy, this platform may help make DNA-NLS delivery more reliable and more dose-efficient.
The broader implication is clear: if DNA therapeutics are to compete in non-viral delivery, the field will likely need better tools for guiding cargo into the nucleus. DNA-PepTAG adds a promising option to that toolkit.