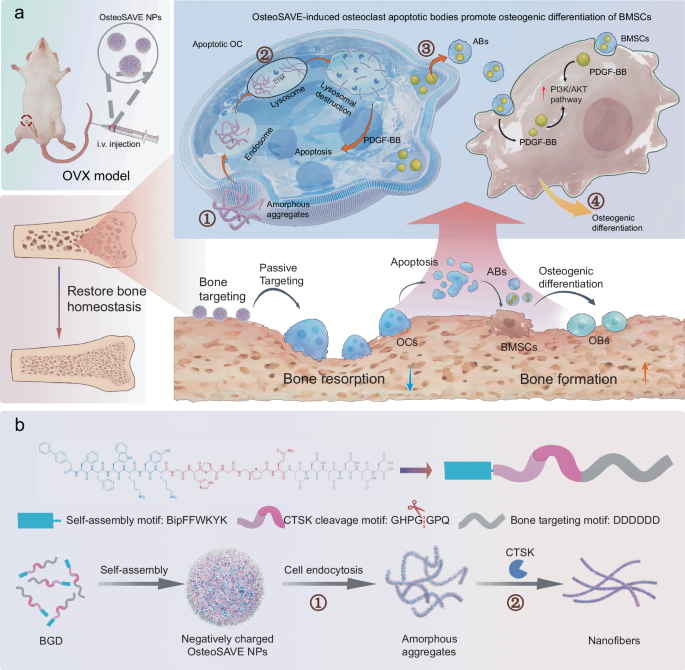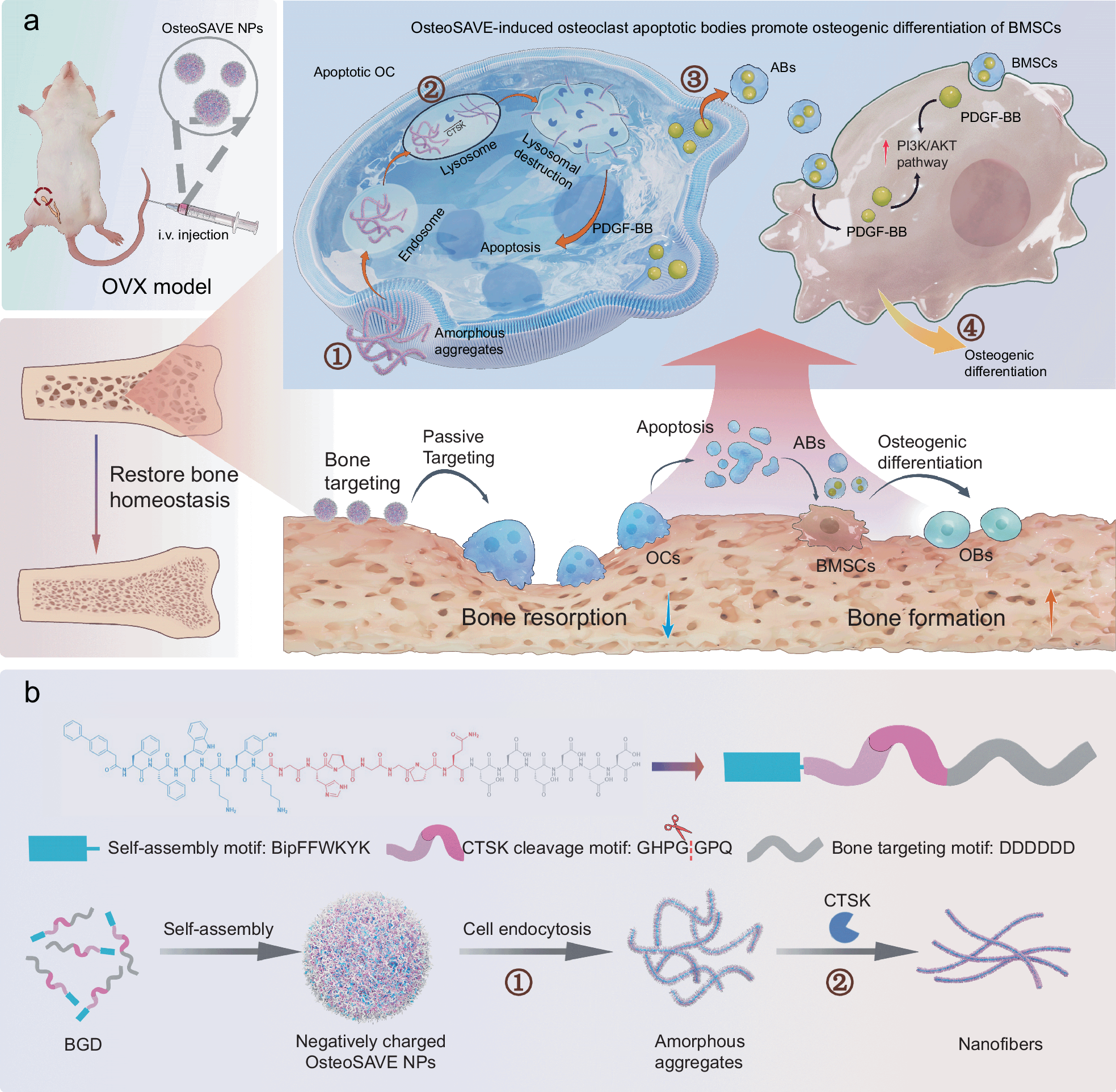
Osteoporosis treatments often face a difficult tradeoff: suppress too much osteoclast activity and normal bone remodeling suffers, but suppress too little and bone loss continues. A new study describes a peptide-based nanoparticle designed to break that stalemate by selectively acting on mature osteoclasts while preserving broader bone turnover.
The system, called OsteoSAVE, combines three functions in one construct: a bone-targeting motif, a cathepsin K-sensitive linker, and a self-assembling core that changes shape after enzyme cleavage. Cathepsin K is highly active in mature osteoclasts, making it a useful biological trigger for distinguishing these cells from earlier precursors.
Once OsteoSAVE is taken up by osteoclast lysosomes, cathepsin K cuts the linker and the particles reorganize into nanofibers. That transformation helps push the mature osteoclasts into apoptosis, and the dying cells release apoptotic bodies enriched with factors such as PDGF-BB. Rather than being just cellular debris, these vesicle-like bodies appear to carry regenerative signals.
In cell and mouse experiments, the resulting apoptotic bodies promoted osteogenic behavior in mesenchymal stem cells and helped restore bone density in an ovariectomized osteoporosis model. The study links this effect to PI3K/AKT signaling, suggesting a pathway through which the osteoclast-derived bodies encourage bone formation.
The approach is notable because it does more than simply block resorption. It aims to remove pathological osteoclasts with precision while simultaneously turning that cell death into a source of pro-bone signals. If translated successfully, this kind of enzyme-responsive supramolecular therapy could point toward a more balanced way to treat osteoporosis.
As a platform, OsteoSAVE highlights an emerging idea in peptide and nanomedicine research: use disease-specific enzymes not only as biomarkers, but as switches that activate therapy exactly where it is needed.