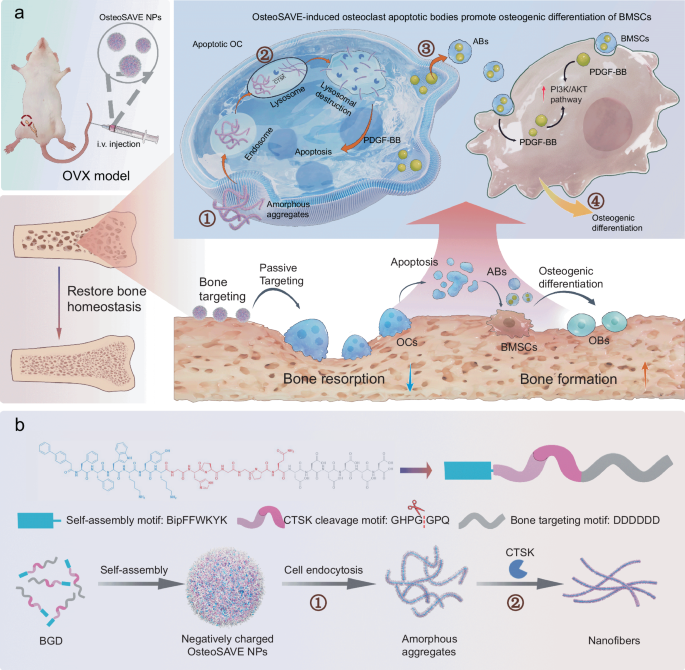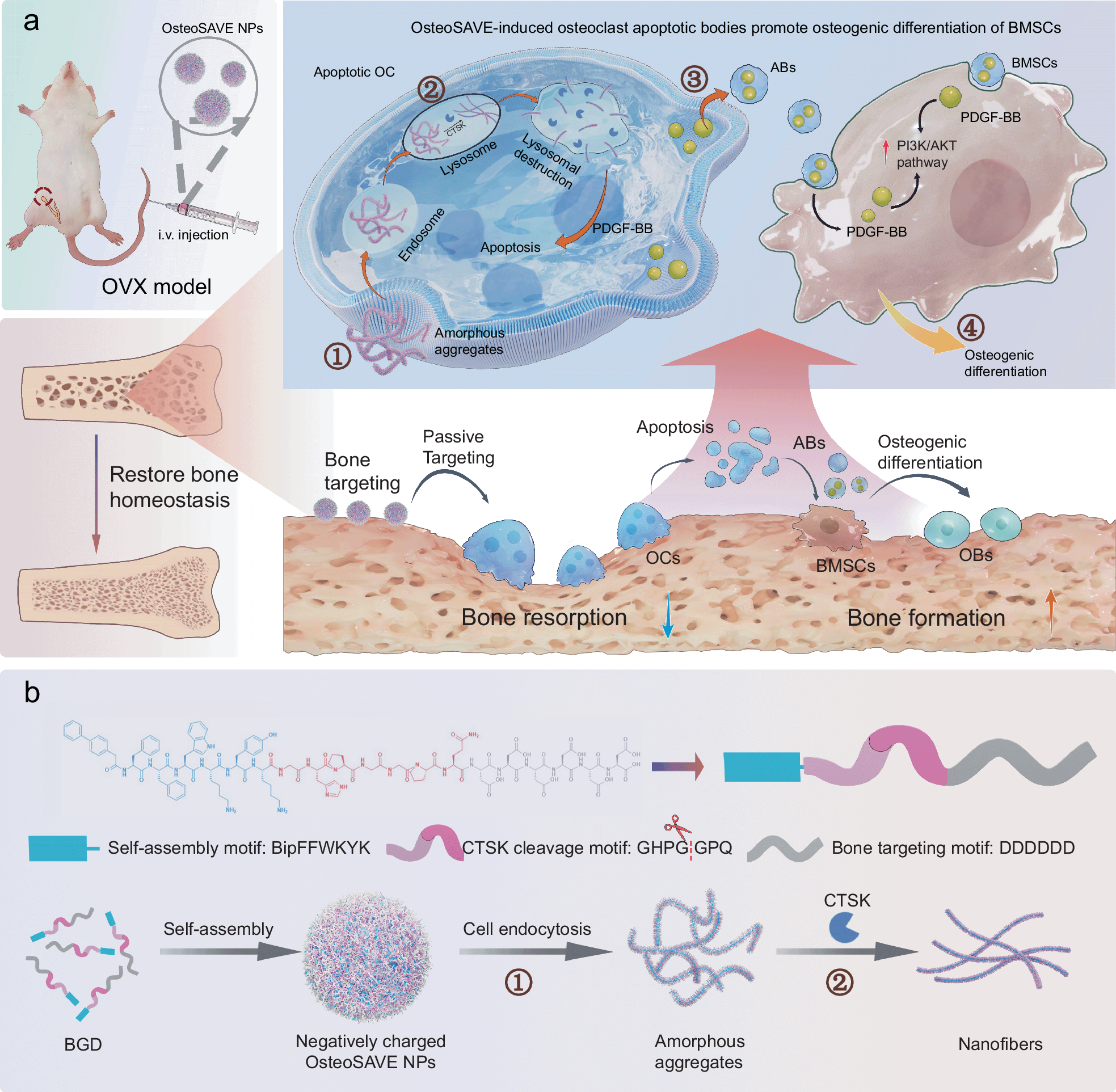
Osteoporosis treatments have long faced a difficult tradeoff: shutting down too much bone resorption can protect against bone loss, but broad inhibition of osteoclasts can also interfere with normal remodeling and raise the risk of complications. A new study points to a more selective approach built around a peptide-based self-assembling nanoparticle called OsteoSAVE.
The system is designed to home in on bone and respond to cathepsin K (CTSK), an enzyme highly active in mature osteoclasts. Once OsteoSAVE is taken up into these cells, CTSK cleaves a linker in the construct and drives a structural shift into nanofibers inside lysosomes. That transformation appears to trigger apoptosis in the mature osteoclasts rather than broadly suppressing bone-resorbing cells across the board.
The interesting twist is what happens next. As the targeted osteoclasts die, they release apoptotic bodies enriched with PDGF-BB. In the study, these vesicle-like fragments helped push mesenchymal stem cells toward osteogenic activity, supporting bone regeneration through activation of the PI3K/AKT pathway.
In ovariectomized mice, a commonly used model of postmenopausal bone loss, OsteoSAVE improved bone density and shifted the balance of bone homeostasis in a favorable direction. The result is a dual-action concept: remove pathologically active osteoclasts while simultaneously generating pro-regenerative signals from their apoptotic remnants.
From a peptide and materials perspective, the design is notable for combining three functions in one platform: a bone-targeting element, an enzyme-responsive cleavage site, and a self-assembling core. That combination is what gives the nanoparticle its selectivity and its in situ remodeling effect.
If translated further, this strategy could represent a new direction for osteoporosis therapy: not just blocking bone breakdown, but using disease-associated biology to trigger a constructive response in the bone microenvironment.