Targeted protein degradation has become one of the most closely watched strategies in chemical biology, but most of the field still depends on recruiting the cell’s own ubiquitination machinery. That dependence can make degrader design tricky, since success often hinges on forming the right E3 ligase-target complex with the right shape, stability, and geometry.
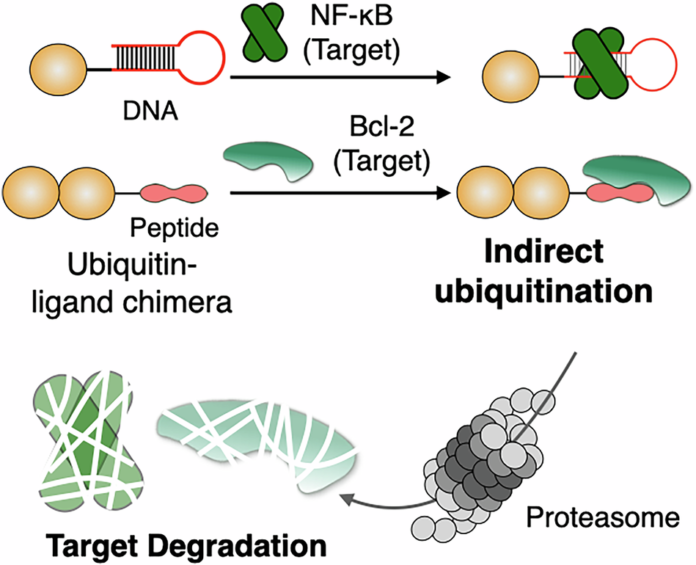
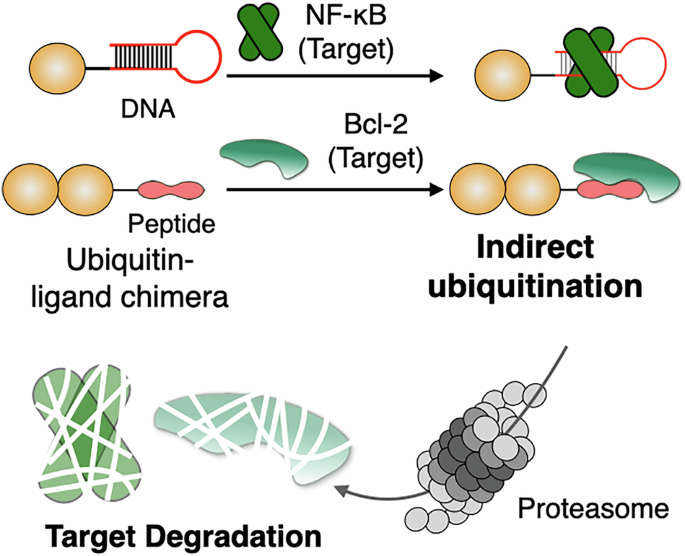
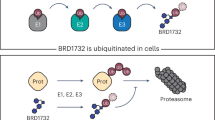
In a new study, researchers describe indirect ubiquitination, a chemical approach designed to bypass endogenous E3 ligases altogether. Their system uses a chimeric molecule built from two parts: a ligand that recognizes the target protein and a ubiquitin component that can be non-covalently positioned so the target receives a ubiquitin tag without the usual E3-mediated handoff.
The result is a form of ubiquitin-based degradation that does not rely on the target’s interaction with a native ligase. In cellular and biochemical testing, the strategy promoted proteasome-dependent degradation of recombinant Bcl-2 and NF-κB p50, and also reduced levels of endogenous Bcl-2 inside cells.
This matters because classic PROTAC-style degraders can run into several practical barriers:
- they may need extensive optimization to achieve productive ternary complex formation,
- their activity can vary with the availability and behavior of endogenous E3 ligases,
- and resistance can emerge through changes in ligase function or at the target-ligase interface.
By moving ubiquitin delivery upstream of endogenous E3 biology, indirect ubiquitination could open a new design space for degrader development. It may also broaden the range of proteins that can be targeted, especially cases where conventional E3-recruiting approaches have struggled.
For peptide and protein researchers, the concept is especially interesting because it reframes ubiquitin not just as a downstream signal, but as a modular chemical payload that can be tethered to a chosen target through binding interactions alone.
The work is still at an early stage, but it adds a fresh tool to the growing degradation toolbox and suggests that non-covalent ubiquitin delivery could become a useful platform for future therapeutics and probe design.