
Pseudomonas aeruginosa is known for thriving in tough environments and for resisting many antibiotics, which makes it a major concern in healthcare settings. One reason for its adaptability is its ability to scavenge nutrients efficiently, including small peptides released during protein breakdown.
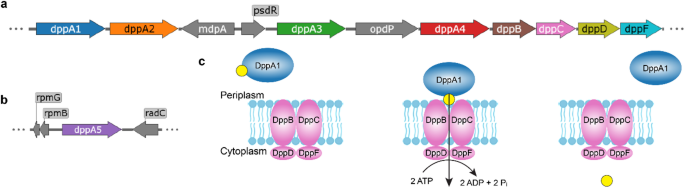
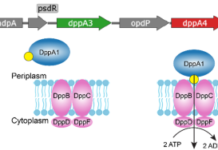
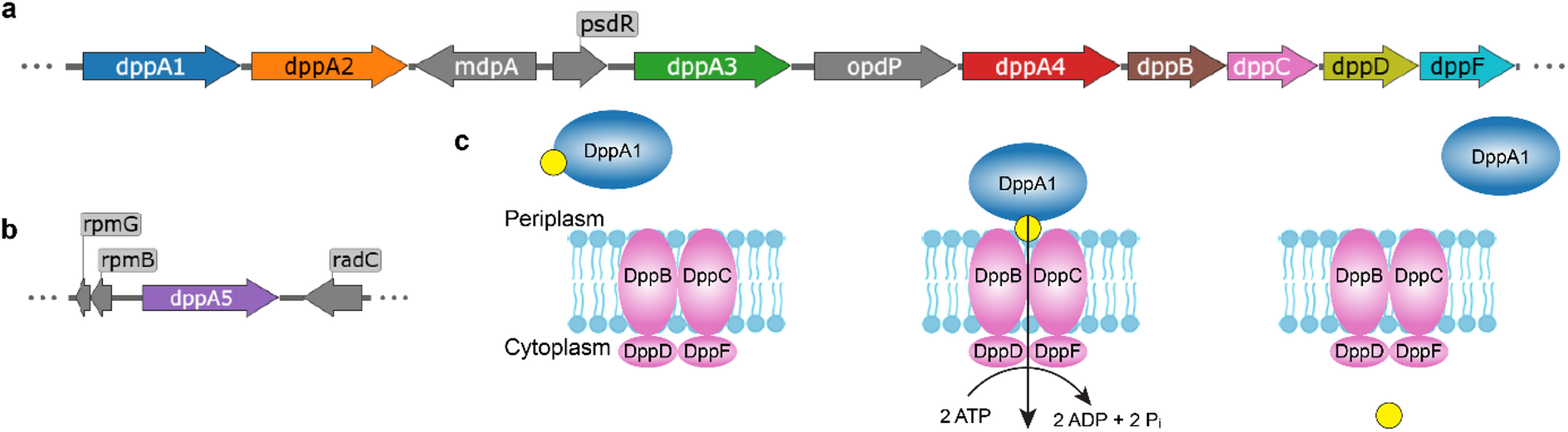
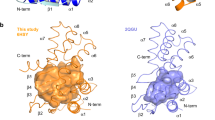
A new study takes a closer look at the bacterium’s dipeptide permease, or Dpp, a peptide transport system that moves short peptides into the cell. Unlike many bacteria that rely on a single peptide-binding protein for this task, P. aeruginosa carries five related DppA proteins. Until now, it was not clear why the organism kept so many versions of what seemed like the same component.
To answer that question, the researchers tested all five DppA proteins against a large panel of 281 dipeptides and tripeptides using Differential Scanning Fluorimetry, a method that can reveal when a ligand stabilizes a protein. The results show that the paralogs are not redundant.
Two of the proteins, DppA1 and DppA3, were found to prefer dipeptides, while DppA2 and DppA4 showed stronger binding to tripeptides. DppA5, however, did not show detectable binding in the assay, hinting that it may have diverged into a different role or requires conditions not captured in the screen.
The findings suggest that the multiple DppA proteins broaden the range of peptide substrates that P. aeruginosa can capture from its surroundings. That could give the pathogen an edge in nutrient-poor or competitive environments, and it may also matter for processes beyond feeding, since peptide-binding systems in bacteria can participate in signaling, chemotaxis, and biofilm-related behavior.
More broadly, the work reinforces a growing idea in microbiology: proteins that look similar on paper may have very different functions in practice. In a pathogen as adaptable as P. aeruginosa, that specialization could help explain both its environmental resilience and its clinical success.
The study also points to possible long-term applications. Because peptide transport systems can be exploited for drug entry, understanding DppA specificity may help inform future Trojan Horse antimicrobial strategies that use peptide-like carriers to smuggle drugs into bacterial cells.