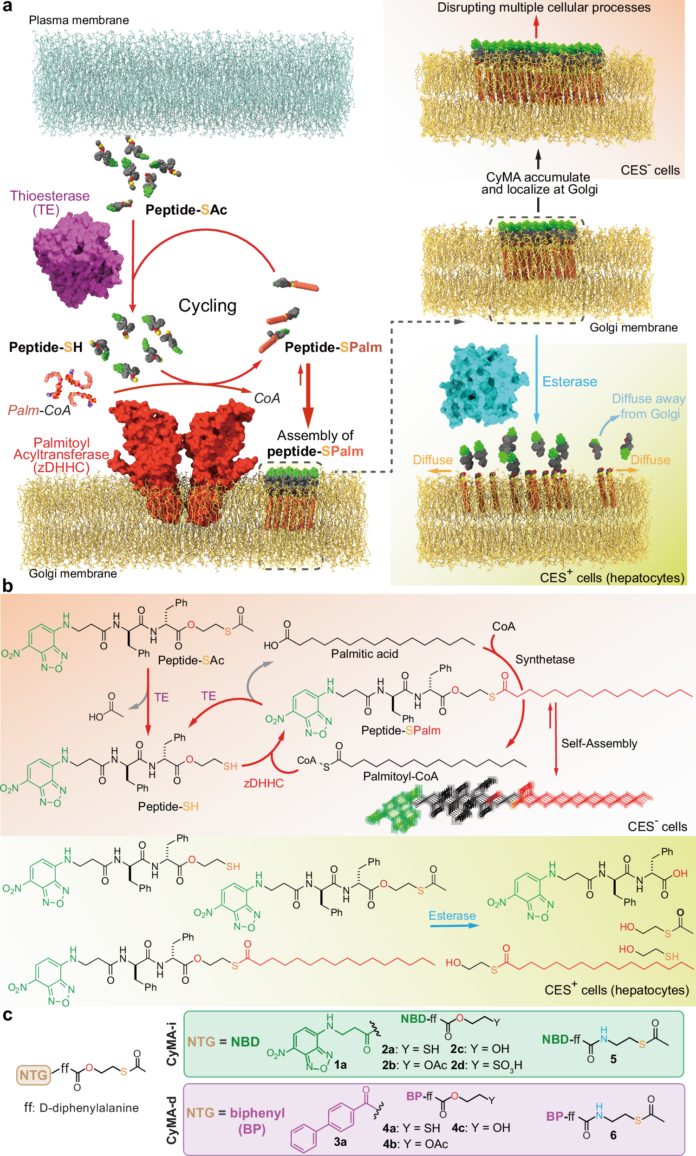

The Golgi apparatus is one of the cell’s busiest sorting and processing centers, but it has been difficult to label quickly or interfere with in a selective way. A new peptide-based strategy called cycling molecular assemblies, or CyMA, aims to change that by using the cell’s own lipidation machinery to trap supramolecular structures at the Golgi.
The approach begins with acetylated, amphiphilic thiopeptide precursors that can cross the plasma membrane. Once inside the cell, thioesterases remove the acetyl group to reveal thiols. Those thiols are then palmitoylated by Golgi-resident acyltransferases, creating palmitoylated peptides that self-assemble into dynamic nanostructures at the Golgi.
Because the palmitoylation and depalmitoylation reactions keep cycling, the assemblies accumulate rapidly and support near-instant Golgi imaging at very low probe levels. In other words, the Golgi is not just labeled passively; it is continuously fed by a futile enzymatic cycle that maintains localization.
The same design can be turned from a reporter into a disruptor. When the fluorescent group is replaced with a biphenyl motif, the resulting CyMA variant builds up at the Golgi and interferes with key functions such as protein modification, trafficking, and secretion. That disruption can mislocalize important proteins, impair receptor signaling, and ultimately suppress cell proliferation.
The study also reports an element of cell selectivity, with some cell types, including hepatocytes and certain immune cells, being less affected. The authors suggest that dynamic supramolecular assembly driven by enzyme switching could be adapted to other organelles by choosing different biochemical “switches” that enable kinetic trapping.