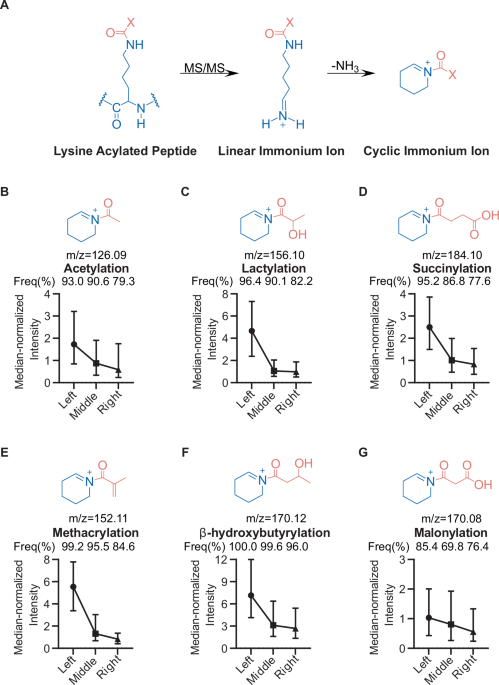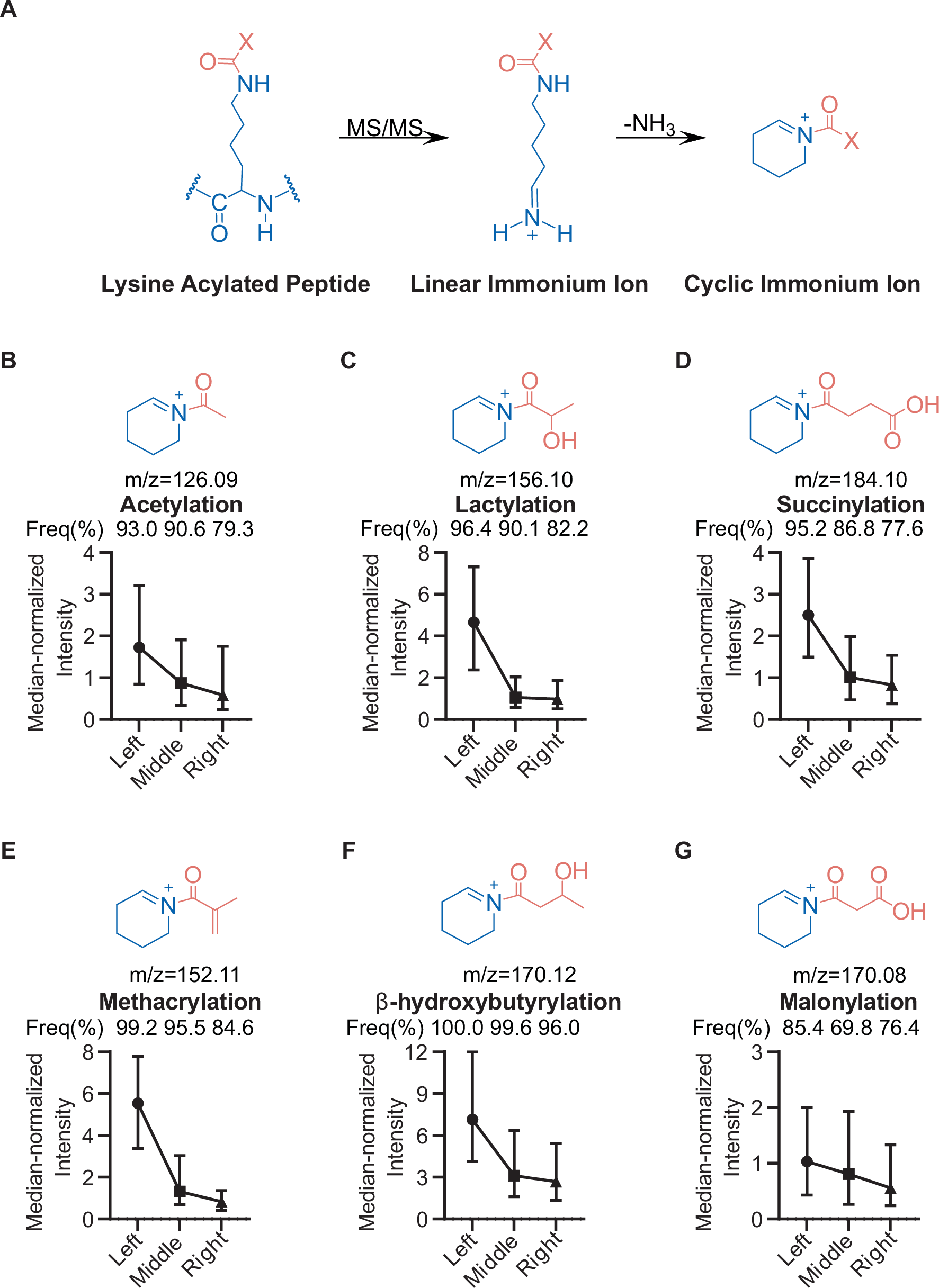
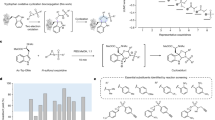
Mass spectrometry remains one of the most powerful tools for mapping protein lysine acylations, but confident site assignment still depends on getting the right diagnostic signals from peptide fragmentation. In a new study, researchers examined one of the most useful markers for acylated lysine: cyclic immonium ions.
The team found that these ions are not produced uniformly across a peptide. Instead, there is a clear positional bias: acylated lysine residues near the N-terminus of a tryptic peptide are much more likely to generate the diagnostic signal. To probe the effect more directly, the researchers used alternative digestion strategies and isotope-labeled synthetic peptides, supporting the idea that the behavior is tied to how b-type fragments break down during collision-induced dissociation.
That insight led to a practical solution. By using stepped higher-energy collision dissociation, the authors improved peptide sequence coverage and boosted cyclic immonium ion detection to nearly complete levels. The result is a faster and more reliable workflow for identifying protein acylations, with higher confidence in both detection and localization.
For proteomics labs studying acetylation, succinylation, crotonylation, and related modifications, the work offers both a mechanistic explanation and an acquisition strategy that could strengthen acylome profiling across complex samples.