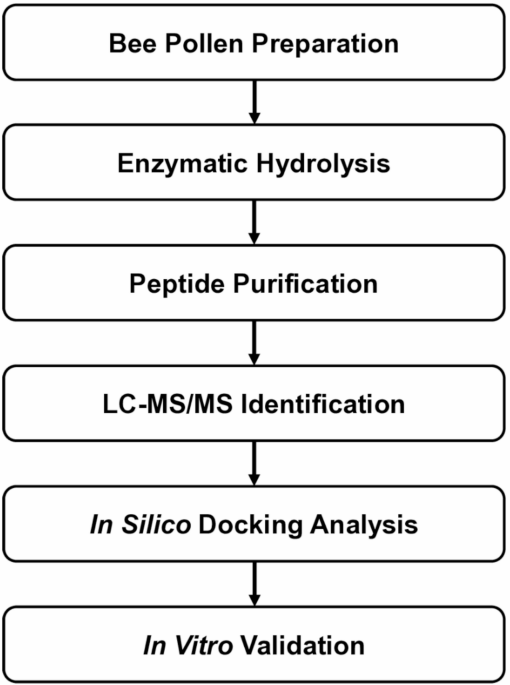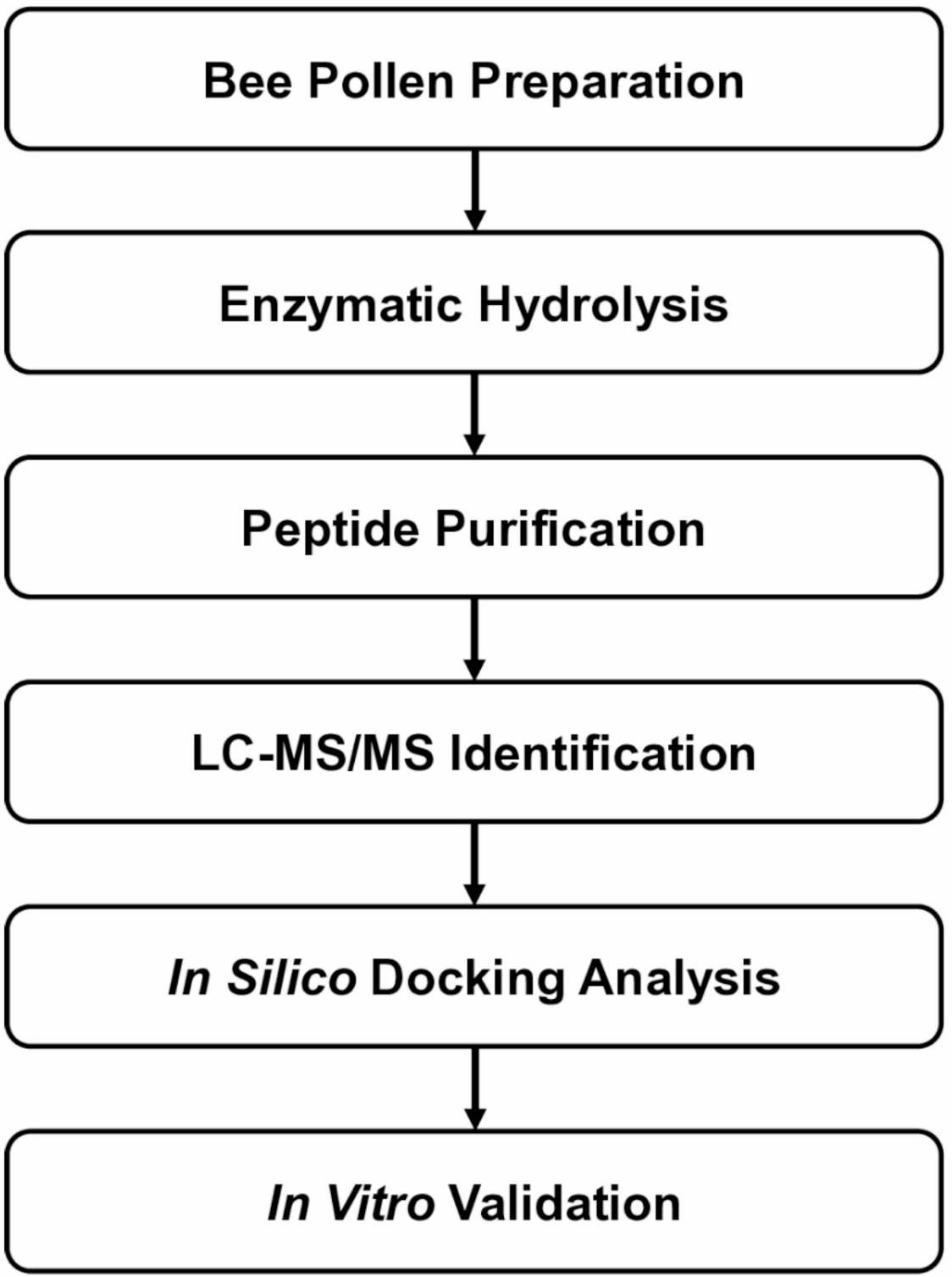
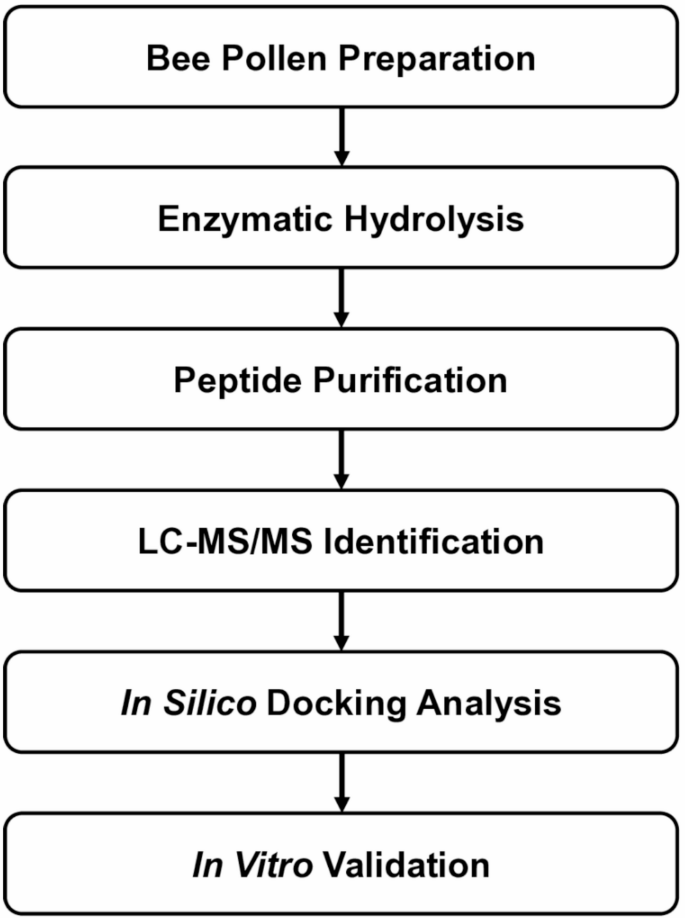
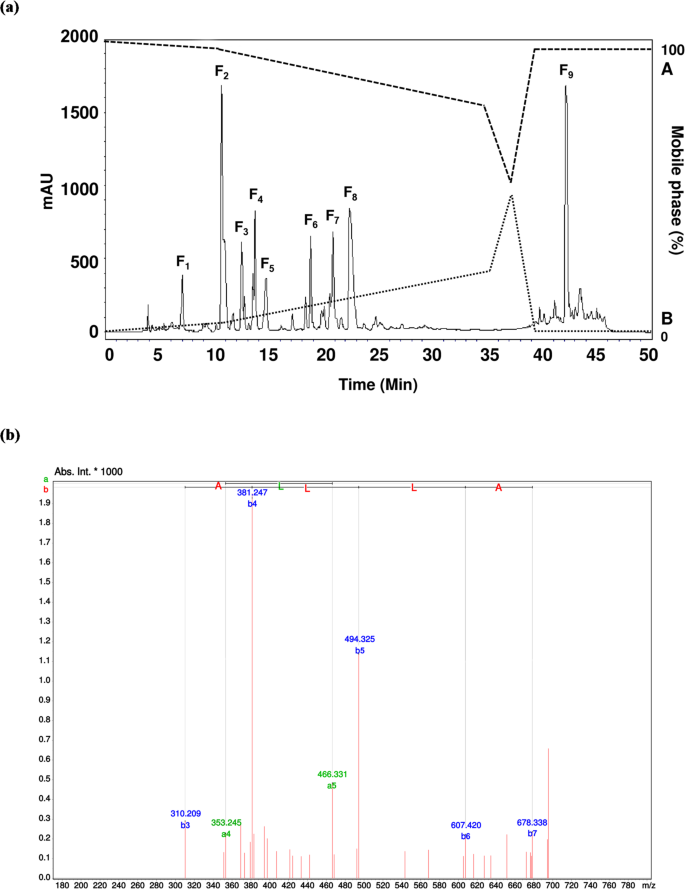
A new study points to bee pollen as a source of bioactive peptides with potential relevance to blood sugar regulation. After enzymatic digestion and purification of bee pollen protein hydrolysates, researchers isolated a seven-amino-acid peptide, Ala-Thr-His-Ala-Leu-Leu-Ala, or ATHALLA (AA-7), that showed two notable activities: inhibition of dipeptidyl peptidase IV (DPP-IV) and modulation of glucose transport-related genes.
DPP-IV is an important target in type 2 diabetes research because the enzyme rapidly breaks down incretin hormones such as GLP-1. By slowing DPP-IV activity, it may be possible to prolong endogenous GLP-1 signaling and support glucose control. In this work, AA-7 inhibited DPP-IV with an IC50 of 52.63 ± 2.32 µM. That places it below the potency of the reference inhibitor diprotin A, but still within a range that supports interest in natural peptide leads.
To understand how the peptide might interact with the enzyme, the team performed molecular docking studies. The modeling suggested that AA-7 fits stably within the catalytic pocket of DPP-IV, with hydrogen bonding and hydrophobic contacts helping anchor the peptide to key residues. While docking does not prove biological function on its own, it provides a structural rationale for the observed inhibition.
The study also looked beyond DPP-IV. In Caco-2 cells, AA-7 influenced the expression of SGLT1 and GLUT2, two proteins involved in intestinal glucose handling. The response was dose-dependent, suggesting that the peptide may affect glucose transport as well as incretin degradation. Additional docking work with SGLT1 and GLUT2 offered further support for possible interactions, though the authors note that these results are best viewed as hypothesis-generating rather than definitive mechanistic proof.
Safety and drug-likeness were also examined using in silico ADMET analysis. The peptide was predicted to have poor passive permeability and limited intestinal absorption, which could complicate oral delivery. At the same time, the computational profile suggested minimal CYP450 interactions and low toxicity, pointing to a potentially favorable safety profile if delivery challenges can be addressed.
Overall, the findings add to growing interest in food-derived peptides as candidates for metabolic health applications. Bee pollen protein hydrolysates may contain sequences that combine enzyme inhibition with effects on glucose transport, opening a possible path toward nutraceutical or functional food development. For now, AA-7 looks like a promising lead rather than a finished therapy, but it highlights how natural products continue to supply new ideas for diabetes research.