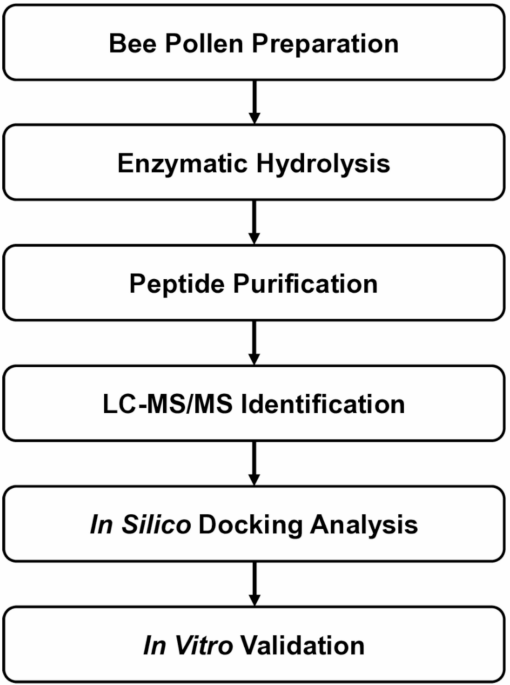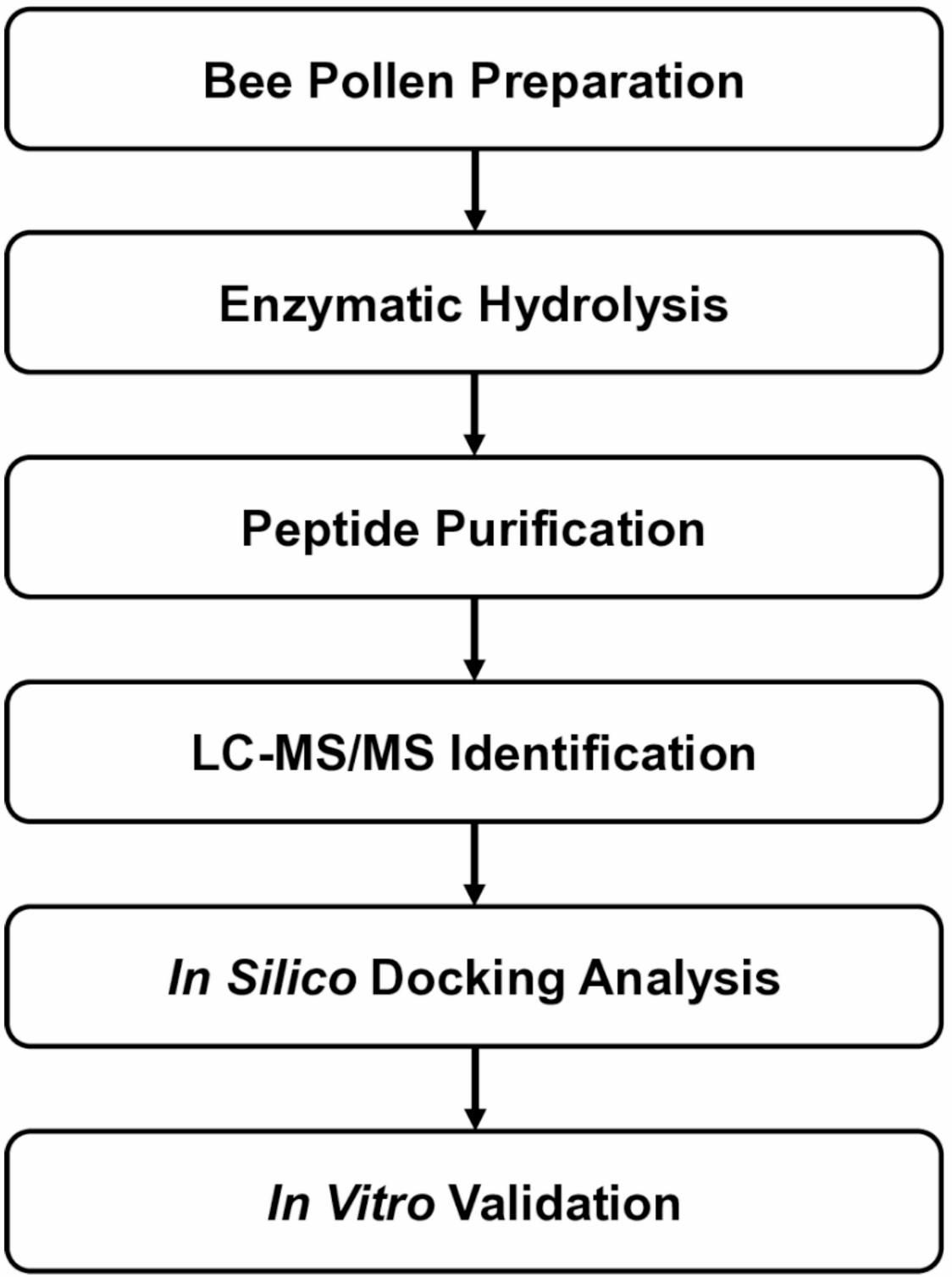
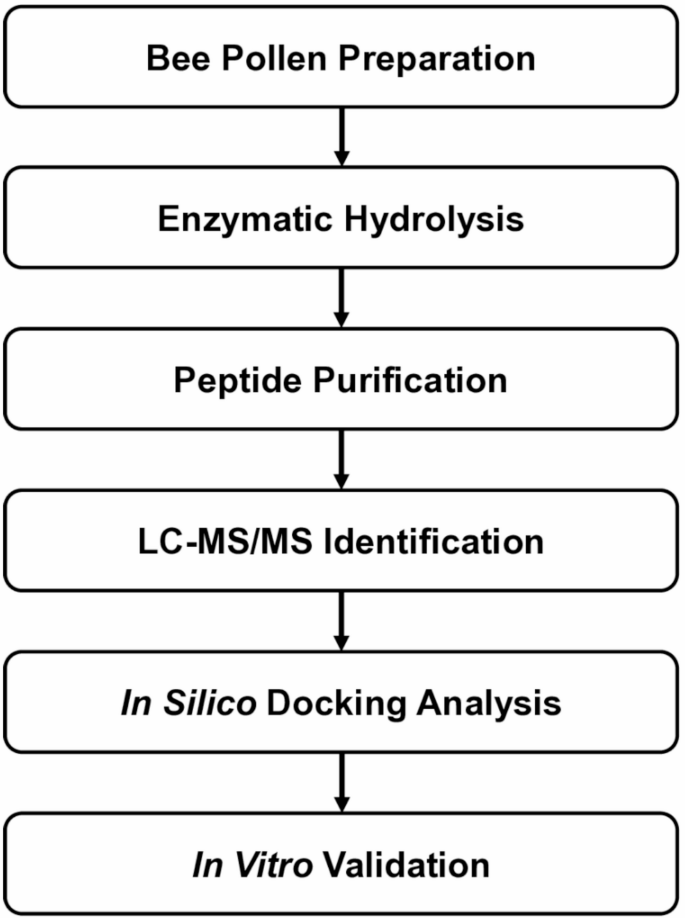
Bee pollen is best known as a nutrient-rich natural product, but a new study suggests it may also contain peptides with activity relevant to type 2 diabetes research. Scientists working with a bee pollen protein hydrolysate isolated a short peptide, ATHALLA (also called AA-7), that showed two notable properties in laboratory experiments: it inhibited the enzyme DPP-IV and it altered the expression of glucose transport genes in intestinal cells.
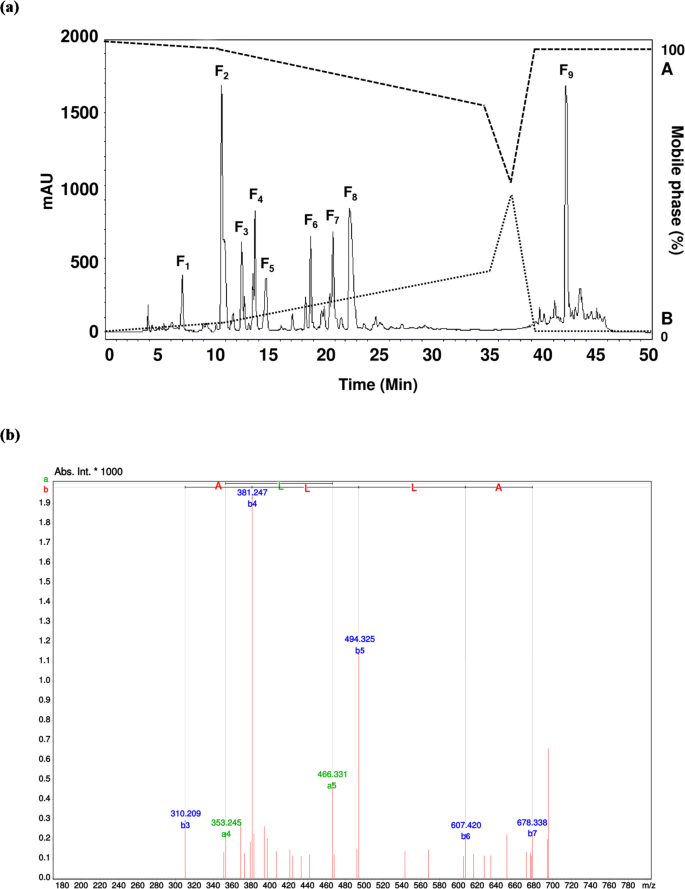
The team produced the hydrolysate using a simulated digestive process with pepsin and pancreatin, then separated peptide fractions with ultrafiltration and purification by reverse-phase HPLC. Using mass spectrometry, they identified ATHALLA as a leading candidate linked to the observed DPP-IV inhibitory activity.
Why DPP-IV matters
DPP-IV is an important target in diabetes research because it breaks down incretin hormones such as GLP-1, which help regulate blood sugar after meals. By slowing DPP-IV activity, it may be possible to extend the action of these hormones and support better glucose control. In this study, ATHALLA inhibited DPP-IV with an IC50 of 52.63 µM. That is weaker than the reference compound diprotin A, but still significant for a naturally derived peptide.
Computational docking backed up the biochemical data, suggesting that ATHALLA can fit into the DPP-IV active site and form a network of hydrogen-bond and hydrophobic interactions with key residues in the enzyme’s catalytic pocket.
Effects on glucose transport in intestinal cells
The study also examined the peptide in Caco-2 cells, a widely used intestinal cell model. Here, ATHALLA appeared to influence genes involved in glucose uptake, including SGLT1 and GLUT2, in a dose-dependent manner. The authors also used docking analyses to explore possible interactions with these transport-related proteins, although they caution that these simulations are supportive rather than proof of mechanism.
In practical terms, this gives the peptide a dual profile: it may act both on hormone regulation through DPP-IV inhibition and on intestinal glucose handling through transporter modulation.
What the early safety and ADMET results suggest
In silico ADMET screening pointed to a few likely hurdles. The peptide was predicted to have poor passive membrane permeability and limited intestinal absorption, which could affect oral bioavailability. On the other hand, it showed minimal predicted CYP450 interactions and low toxicity signals, which is encouraging from a safety perspective.
That combination is common in early peptide research: strong biological activity in vitro, but pharmacokinetic challenges that may require formulation strategies or peptide optimization before real-world use becomes plausible.
Why this matters for nutraceutical research
The findings add to a growing body of work suggesting that food-derived peptides can do more than provide nutrition. If further studies confirm the activity and improve delivery, bee pollen peptides could become candidates for functional foods or nutraceuticals aimed at glycemic support.
For now, the work should be viewed as an early-stage discovery: promising in enzyme assays, supported by molecular modeling, and active in a cell model, but not yet tested in animals or humans. Still, ATHALLA stands out as a useful lead for researchers exploring natural DPP-IV inhibitors with added effects on glucose transport.