A new report in Nature Sustainability describes a solid-phase peptide synthesis (SPPS) strategy designed to reduce reliance on traditional organic solvents. The method uses a hydrophilic, biodegradable poly-ε-lysine-based solid support and carries out the synthesis in aqueous solution.
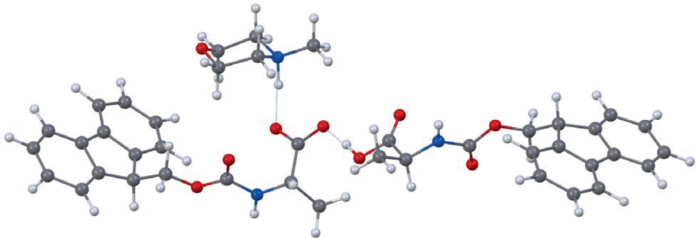
The key enabling step is the preparation of Nα-Fmoc-protected amino acids as water-soluble salts. According to the authors, this can be achieved by pairing the amino acids with either N-methylmorpholine (NMM) or N,N,N-triethanolamine (TEOA), allowing high-concentration aqueous handling of building blocks that are normally difficult to dissolve in water.
Building on earlier observations that Fmoc amino acids become soluble after pre-dissolution with at least one equivalent of NMM, the team shows that the same salification-driven solubility principle extends across 20 standard Fmoc amino acids. That broad compatibility is important for peptide synthesis, where access to a full amino acid toolkit determines how versatile a platform can be.
Structural analysis also helped explain why the approach works. Using 3D electron diffraction, the researchers confirmed a 1:1 stoichiometry in the crystalline amino acid-base complexes and found evidence consistent with deprotonation of the amine components inside those salts.
For peptide chemists, the appeal of the system is clear: water replaces much of the usual solvent burden, and the solid support is both hydrophilic and biodegradable. While the report is still a research-stage advance, it points toward a more sustainable SPPS workflow that could be attractive for future peptide production and process development.