A new report in Nature Sustainability describes a notable shift for solid-phase peptide synthesis: carrying out the chemistry in water rather than relying on traditional organic solvents. The approach uses a hydrophilic, biodegradable poly-ε-lysine-based support, aiming to make peptide assembly cleaner and potentially more sustainable.
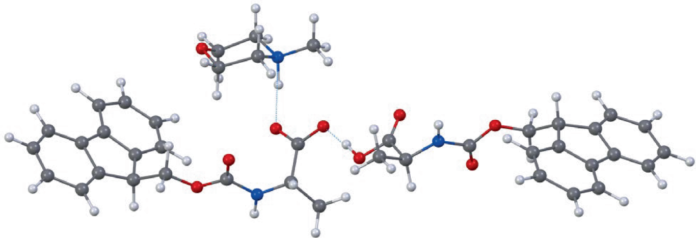
The key challenge in moving peptide synthesis into aqueous media is solubility. Many standard N-α-Fmoc-protected amino acids prefer organic conditions, which has limited how far water-based systems could go. In this work, the team shows that those building blocks can be converted into water-compatible forms by pairing them with bases such as N-methylmorpholine (NMM) or N,N,N-triethanolamine (TEOA). At sufficiently high concentrations, the amino acid salts remain usable in solution, enabling the synthesis workflow to proceed in water.
According to the report, this solubilization strategy was extended across a broad set of 20 conventional Fmoc amino acids. That matters because peptide synthesis platforms are only as useful as the range of residues they can handle. Demonstrating compatibility with the standard amino acid toolkit suggests the method is not just a one-off curiosity, but a potentially practical route for routine peptide assembly.
Structural analysis also helped explain why the method works. 3D electron diffraction of the amino acid-base crystals supported a 1:1 association between the components and indicated deprotonation of the amines within the complexes. In other words, the amino acids are not merely suspended in water; they are chemically reorganized into forms that behave more favorably in solution.
For peptide researchers, the appeal is straightforward: fewer problematic solvents, a biodegradable support, and a solubility trick that may help bring SPPS closer to a more environmentally responsible process. While additional optimization will likely be needed before widespread adoption, the study adds momentum to efforts to make peptide manufacturing less resource-intensive without sacrificing access to standard synthesis chemistry.
If this approach scales well, it could influence how labs think about both sustainability and workflow design in peptide production. Water has long been the ideal reaction medium in principle; this work shows it may be becoming more realistic in practice for solid-phase peptide synthesis.