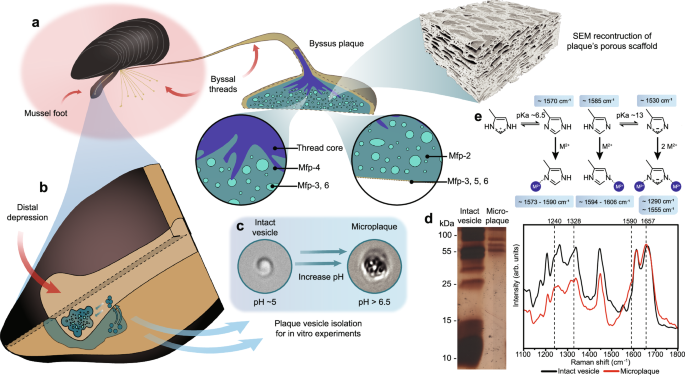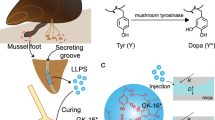

Mussels have long inspired researchers building underwater adhesives, largely because of the famous role of DOPA-containing proteins. But a new study suggests that the picture is broader than a DOPA-only mechanism. The work highlights a previously overlooked histidine-rich plaque protein, mefp-12, as an important contributor to how mussel glue forms, hardens, and achieves its distinctive porous architecture.
Using biochemical localization, the researchers found mefp-12 inside vesicles of the mussel foot’s glue-secreting glands, where adhesive proteins are stored before release. Sequence modeling suggested that the protein can adopt zinc-stabilized coiled-coil structures and contains regions with zinc-finger-like features. That prediction fits with the protein’s unusually high histidine content, which gives it the capacity to respond strongly to metal ions and local pH.
In vitro tests with a histidine-rich α-helical peptide derived from mefp-12 showed a sequence of physical transitions that may mirror what happens in the animal. In the presence of zinc, the peptide underwent liquid-liquid phase separation, forming fluid condensates that spread across surfaces. When exposed to seawater-like pH conditions, those condensates reorganized into solid, nanoporous networks resembling native mussel plaque.
The findings suggest that histidine-mediated metal coordination and pH-triggered self-assembly help drive the curing of mussel glue, not just DOPA chemistry. That shift in emphasis could matter for biomaterials research, since the ability to create wet adhesives that both spread and then set into a porous solid remains a major challenge in synthetic systems.
More broadly, the study adds a new layer to the biology of mussel adhesion: plaque formation appears to depend on a coordinated secretory process, with proteins stored in droplets, released, and then transformed into a functional adhesive structure through environment-sensitive assembly. For engineers, that natural sequence offers a compelling blueprint for next-generation bio-inspired glues.