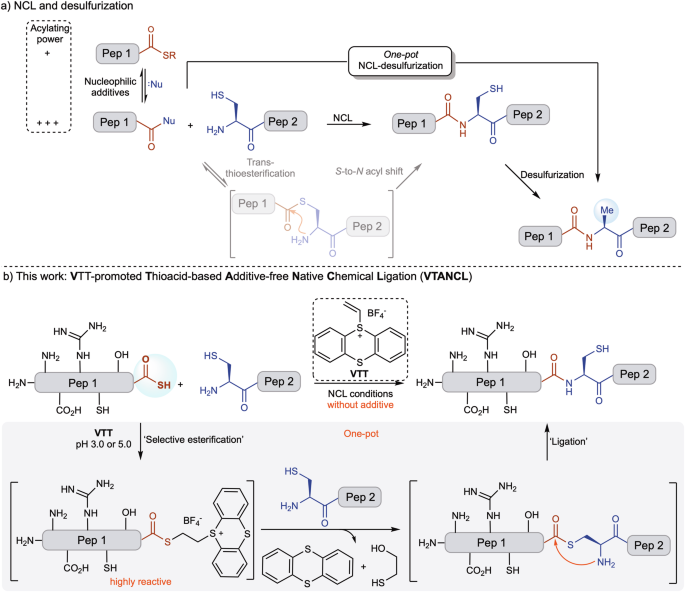
Native chemical ligation (NCL) remains one of the most important tools in chemical protein synthesis, but it has long come with a practical tradeoff: peptide thioesters can be sluggish, so chemists often rely on large amounts of thiol or other nucleophilic additives to keep the reaction moving. Those additives can complicate downstream steps, especially when a one-pot ligation/desulfurization sequence is desired.
In a new study, researchers describe a way around that bottleneck. They show that vinyl thianthrenium tetrafluoroborate (VTT) can rapidly and selectively activate fully unprotected peptide thioacids, converting them into highly reactive thioester intermediates. The result is a fast, additive-free ligation with N-terminal cysteinyl peptides and no detectable epimerization under the reported conditions.
That matters because the classic NCL workflow often depends on excess nucleophilic additives such as MPAA to achieve practical reaction rates. While effective, those additives can interfere with radical-based metal-free desulfurization, forcing researchers to remove them before moving on. By eliminating the additive requirement at the ligation stage, the VTT method is designed to better fit streamlined one-pot protein synthesis.
The authors also report that the strategy is compatible with post-ligation desulfurization, enabling a more efficient sequence in a single vessel. In their demonstrations, they used the method to assemble hyalomin-3 from two fragments through a one-pot thioesterification-ligation-desulfurization process. They also applied it to ubiquitin using a one-pot three-segment condensation workflow, completing the synthesis in six steps without isolating intermediates.
Beyond the specific examples, the broader appeal is clear: faster ligation, fewer additives, less handling, and improved compatibility with one-pot protein assembly. For chemical protein synthesis, especially projects that depend on desulfurization or multisegment assembly, methods like this can reduce complexity while preserving the chemoselectivity that makes NCL so valuable.
As CPS continues to expand into the preparation of proteins with nonstandard amino acids, probes, and tailored modifications, practical improvements to ligation chemistry remain highly relevant. VTT-promoted thioacid ligation adds another useful option to the toolkit, particularly for researchers looking to simplify native chemical ligation workflows without sacrificing efficiency.