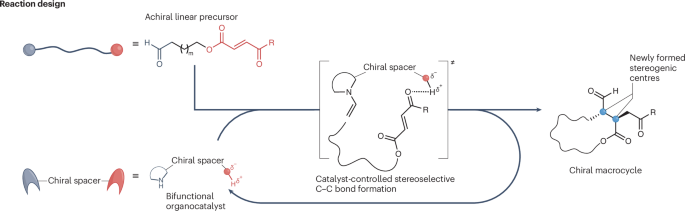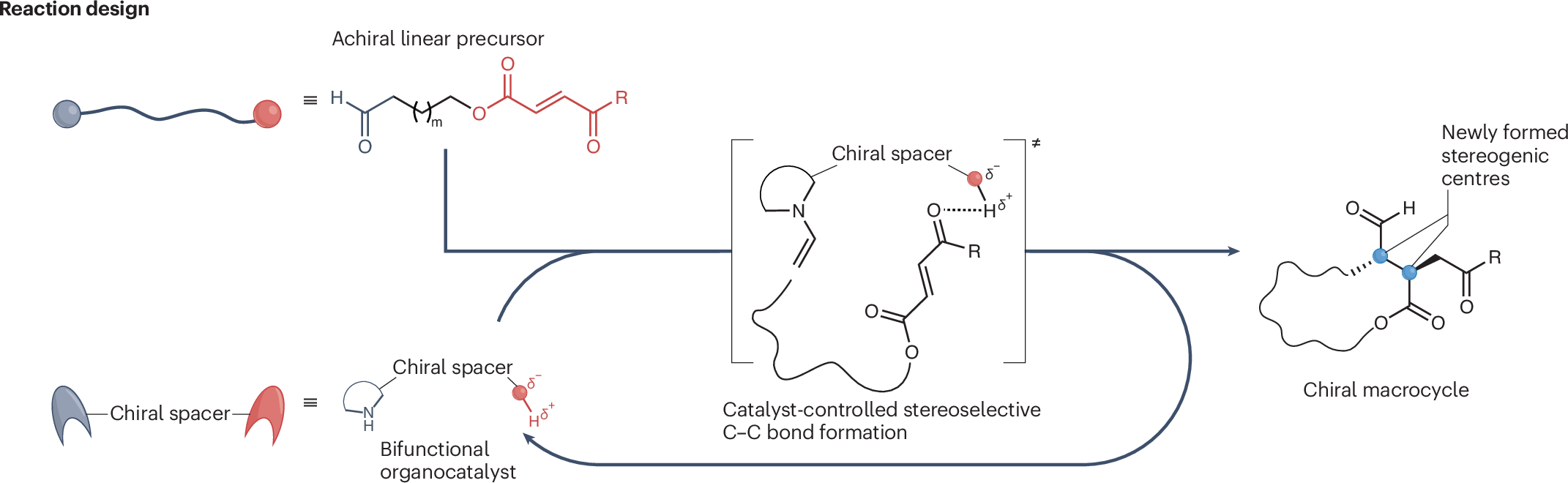
Macrocycles are often prized in medicinal chemistry because their rigid shapes can improve binding, selectivity, and stability. But making them in a controlled way is notoriously difficult, especially when a reaction has to build a large ring while also setting stereochemistry with precision.
A new study in Nature Catalysis describes a peptide-based organocatalyst that solves much of that problem. The catalyst is a tripeptide built from proline-derived elements: D-Pro-α-MePro-Glu-NH2. After screening related proline-inspired catalysts and fine-tuning the reaction conditions, the researchers found this sequence to be the most effective for directing macrocyclization.
What makes the catalyst unusual is its bifunctional design. One part activates the substrate through enamine formation, while another part helps organize the linear precursor into a favorable folded arrangement for ring closure. The glutamic acid side chain appears to play an important preorganization role through noncovalent interactions with the Michael acceptor in the substrate, helping bias the reaction toward intramolecular bond formation rather than unwanted side products.
Using this strategy, the team converted linear substrates containing 12- to 18-membered ring targets with excellent results. Reported selectivity reached 20:1 diastereoselectivity and 99% enantiomeric excess, while macrocycle formation dominated over dimerization in most cases. Computational work supported the proposed stereocontrol model and also helped explain why smaller rings are disfavored: the energetic cost of forming rings below 12 members is too high.
The scope was not limited to one type of Michael acceptor. The researchers also showed that similar outcomes could be obtained with other acceptor motifs, including enamides. Importantly, changes to the linear backbone, including the presence of sp2– and sp-hybridized carbons, did not dramatically erode performance. Even when backbone stereocenters were present, the catalyst remained the main source of stereochemical control.
The method was further demonstrated in late-stage functionalization at an aldehyde handle, and it was applied to the synthesis of a macrocyclic core related to robotnikinin, a molecule of therapeutic interest because of its interaction with the Sonic Hedgehog pathway.
Overall, the work suggests that a well-designed peptide organocatalyst can do more than accelerate a reaction: it can actively choreograph macrocycle formation with predictive stereocontrol. That could make this approach useful for building macrocycles and macrolide-like scaffolds with potential biomedical value.