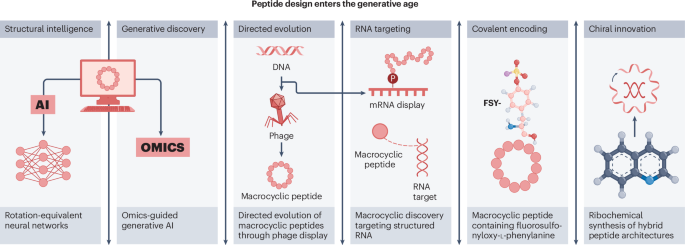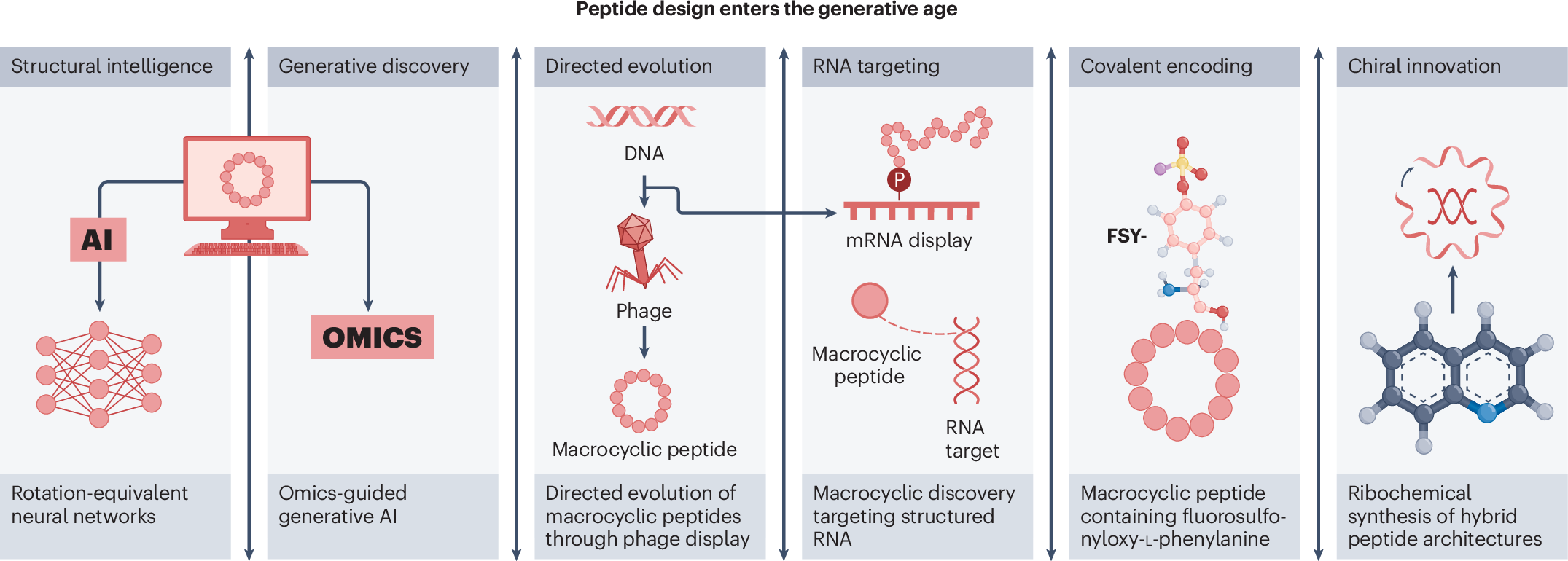
Peptide research in 2025 is increasingly defined by integration rather than isolation. Instead of relying only on stepwise analogue generation, the field is now blending synthetic chemistry with computational design to create molecules that are more selective, more structurally elaborate, and more useful as functional tools.
That shift matters because peptides have long sat at an interesting intersection: they can be built with the precision of chemistry, yet they can still behave like biologically active molecules with rich recognition properties. In the newest wave of work, researchers are pushing peptides into new territory by treating them as programmable molecular platforms rather than simply optimized versions of known sequences.
AI is changing the design process
One of the clearest developments is the growing role of artificial intelligence in peptide discovery. Deep-learning approaches are now helping researchers propose macrocycles with strong binding properties, reducing the dependence on purely trial-and-error screening. This does not replace chemistry; instead, it makes chemical synthesis more targeted and efficient.
Computational methods are also being used to search unusual biological sources for peptide leads, including venom-derived molecules with antimicrobial potential. By expanding the search space beyond conventional libraries, these tools can uncover candidates that would be difficult to find through standard workflows.
Macrocycles are becoming more versatile
Macrocyclic peptides continue to stand out as a major design format because their constrained shapes can improve potency, selectivity, and stability. Recent studies point to increasingly sophisticated ways to discover and optimize these molecules, including display-based platforms and de novo design strategies.
New methods are also making macrocycles useful for a wider range of targets. Instead of focusing only on protein binding, researchers are now reporting macrocyclic peptides that can interact with enzymes, structured RNA, and other challenging biological surfaces.
Covalent chemistry is expanding the toolkit
Another important trend is the rise of target-specific covalent binders. By introducing reactive functionality into peptide scaffolds, scientists can create molecules that not only recognize a target but also form a durable chemical link with it. This can strengthen engagement and open the door to new therapeutic and probe applications.
Several 2025 studies illustrate this direction, including work on ribosomally incorporated noncanonical residues and on peptides designed to modify structured RNA. These projects show that covalent reactivity can be engineered into peptides with increasing precision.
Why this moment is different
The bigger story is that peptide chemistry is no longer advancing mainly by adding more analogues to a growing list. The field is becoming more integrated, predictive, and creative, combining:
- machine learning for sequence and structure design
- display technologies for discovery and selection
- noncanonical building blocks for new functionality
- macrocyclic and atropisomeric frameworks for structural control
- covalent mechanisms for durable target engagement
Taken together, these advances suggest that peptides are evolving from biologically inspired fragments into customizable molecular systems. For researchers, that means a broader design space. For drug discovery, it means more routes to difficult targets. And for the field as a whole, it marks a move from accumulation to architectural innovation.