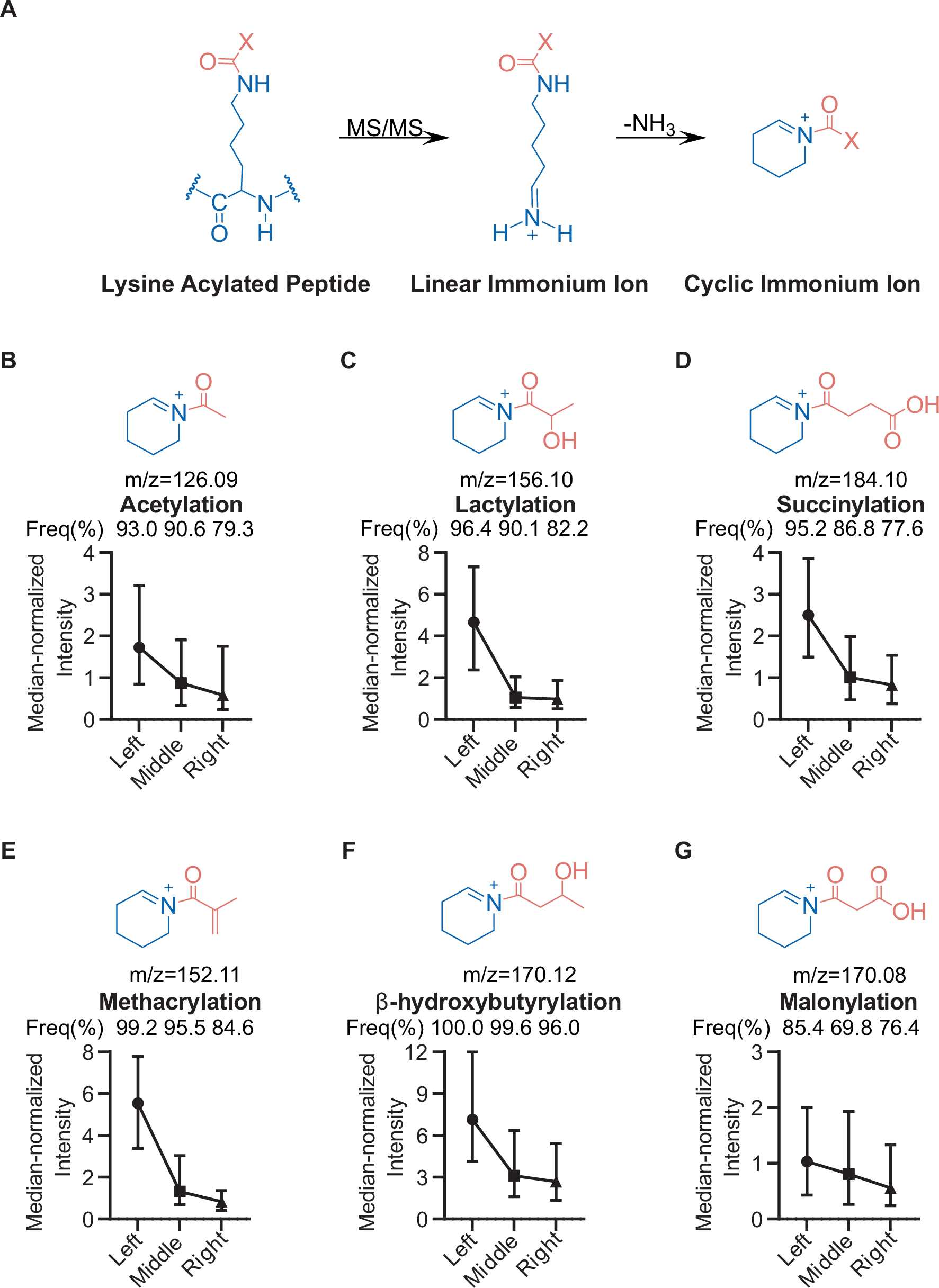
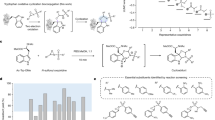
Mass spectrometry has become one of the most important tools for mapping protein lysine acylation, but confident site localization still depends on getting the right fragment ions in the spectrum. A new study highlights an overlooked complication: the diagnostic cyclic immonium ions used to flag lysine acylations are not generated evenly across peptide positions.
The researchers found a strong positional bias. When the modified lysine appears closer to the N-terminus of a tryptic peptide, cyclic immonium ions are much more likely to form. That means the same modification can be easier to detect in one peptide context than another, creating an uneven view of the acylome.
To probe the cause, the team combined LysargiNase digestion with isotope-labeled synthetic peptides. Their results point to the instability of b-type fragment ions during collision-induced dissociation as a major driver of the effect. In other words, the way a peptide breaks apart during MS/MS can favor diagnostic ion formation depending on where the modified residue sits in the sequence.
The practical payoff is a better acquisition strategy. By using stepped higher-energy collision dissociation, the authors report improved sequence coverage and very robust cyclic immonium ion detection, approaching 99% in their workflow. That translates into faster, deeper, and more reliable identification of protein acylation sites.
For proteomics labs studying acetylation, succinylation, crotonylation, lactylation, and related lysine modifications, the message is clear: fragmenting peptides at a single fixed energy may leave blind spots. A stepped-energy approach can help balance the detection of diagnostic ions across different peptide contexts and improve confidence in PTM calls.
Beyond the technical advance, the study also sheds light on a basic question in tandem mass spectrometry: why some diagnostic ions appear readily while others do not. By clarifying how cyclic immonium ions form, the work gives researchers a more dependable route to acylation analysis and a better framework for interpreting spectra.