Solid-phase peptide synthesis remains the workhorse method for making peptides used in research and development, but it comes with a major environmental cost. Conventional protocols rely heavily on organic solvents and generate large amounts of hazardous waste, creating pressure for cleaner alternatives as regulations tighten.
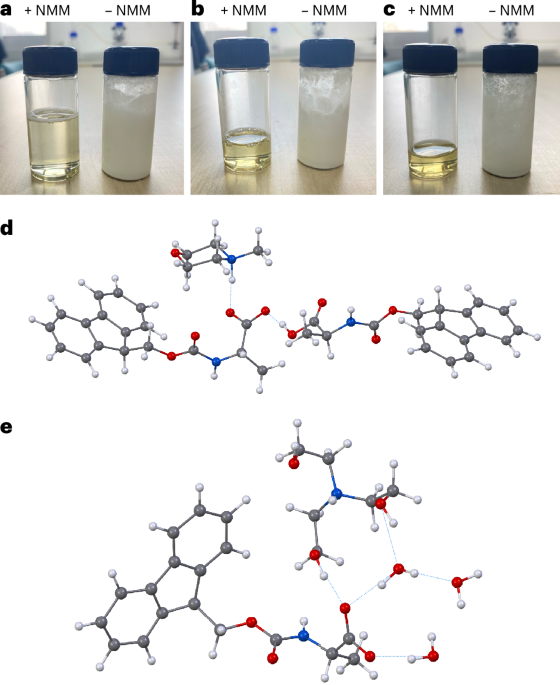
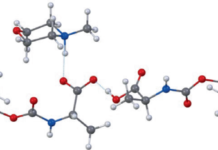
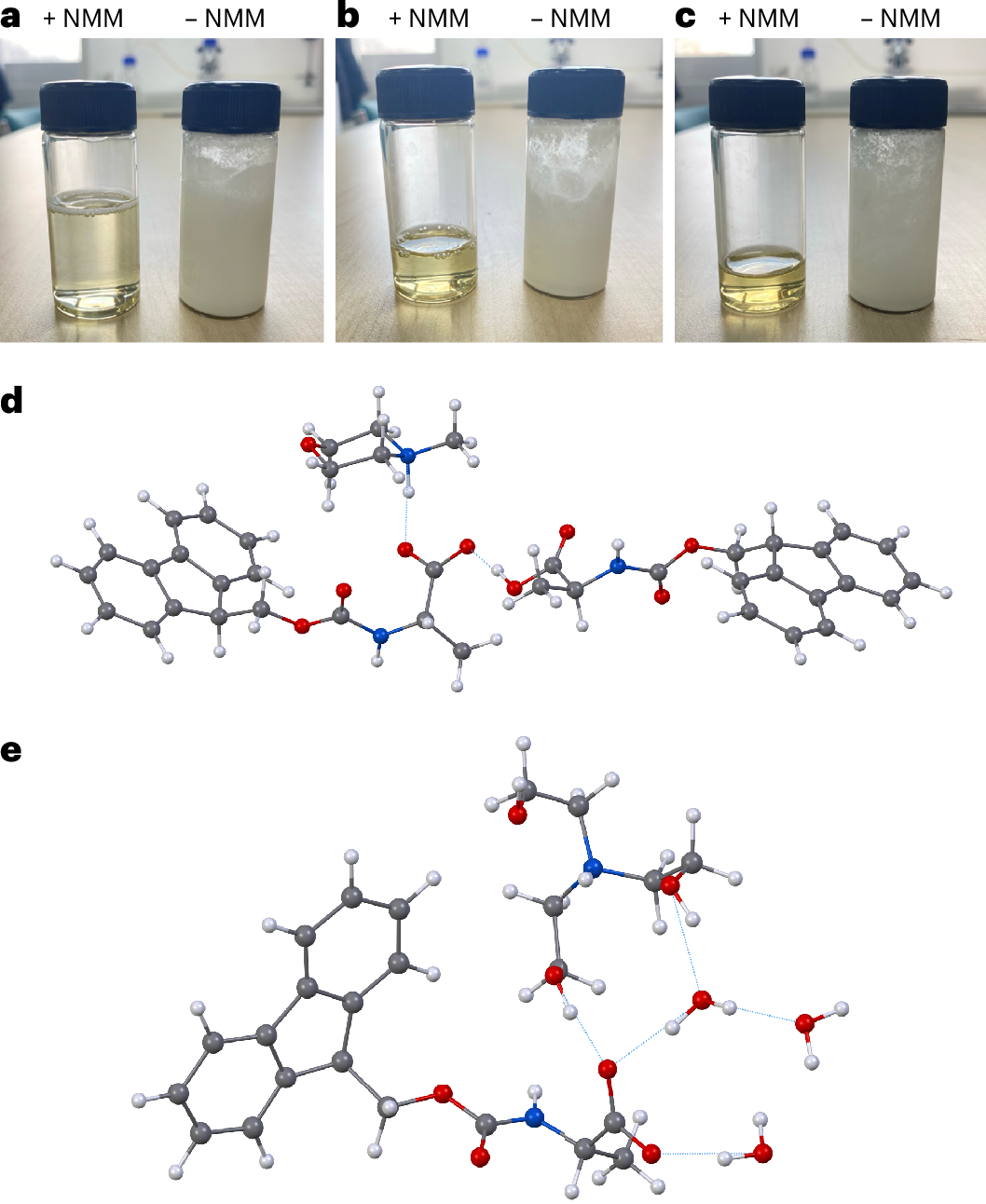
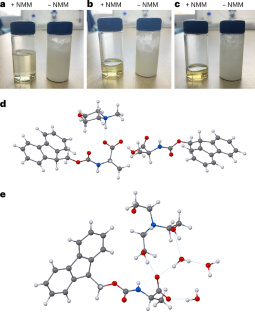
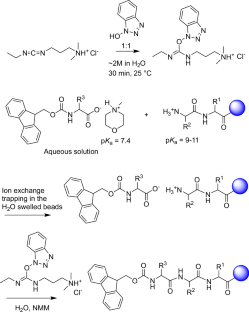
A new study describes a water-based approach that tackles one of the central obstacles to greener peptide chemistry: the poor water solubility of the standard Fmoc-protected amino acids used in SPPS. The researchers found that converting these amino acids into salts with common amines, such as N-methylmorpholine or triethanolamine, can make them highly soluble in water at useful concentrations.
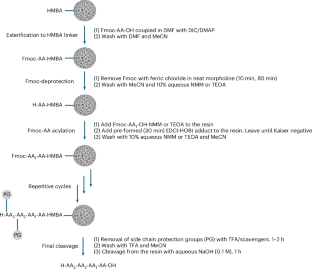
Using a pre-formed, water-soluble activating agent, the team then carried out peptide coupling on a hydrophilic, biodegradable poly-ε-lysine-based solid support. According to the report, the method produced high-quality peptide chains in aqueous conditions while avoiding the side reactions that often complicate synthesis on solid supports.
The significance of the work is not just technical, but practical: peptide manufacturing is increasingly expected to reduce solvent use, and a simpler water-based workflow could help make peptide synthesis more scalable and more environmentally responsible.
If adopted broadly, this kind of approach could reshape how peptide libraries and larger peptide candidates are prepared for applications spanning therapeutics, agriculture, veterinary medicine, and cosmetics.