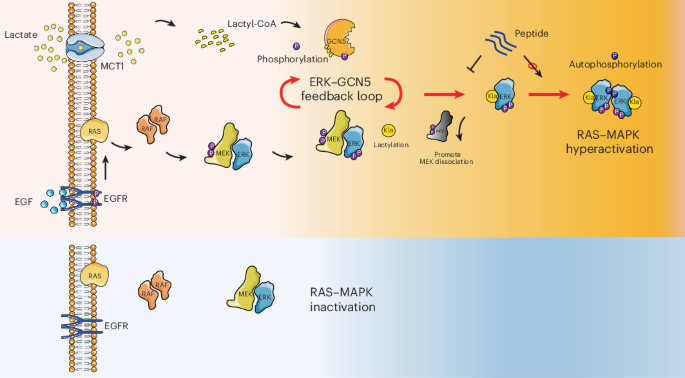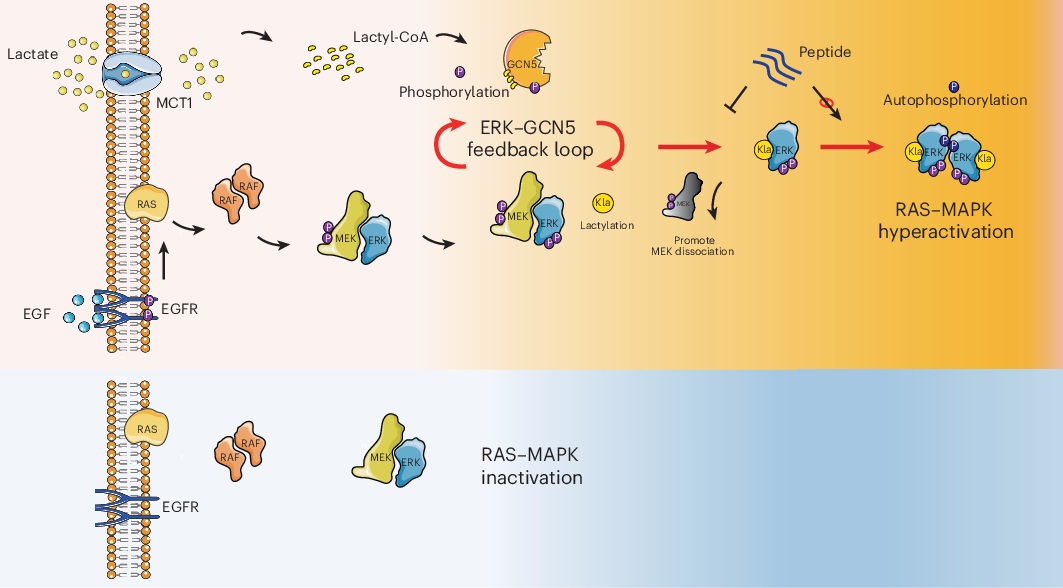
Cancer cells are often described as metabolically rewired, but this study adds a more direct signaling twist: the lactate produced by the Warburg effect does not just mark altered metabolism, it actively helps drive tumor progression.
Researchers report that lactate can switch on the MAPK pathway by promoting a post-translational modification on ERK known as lactylation. In this work, the enzyme GCN5 was identified as the lactyltransferase that installs that mark. Once ERK is activated, it feeds back to phosphorylate GCN5, which in turn boosts GCN5’s ability to lactylate ERK again. The result is a self-reinforcing loop that strengthens lactate-dependent cancer signaling.
How lactylation changes ERK behavior
The key modification was mapped to lysine 231 on ERK. According to the study, lactylation at this site weakens ERK’s interaction with MEK, which appears to favor ERK dimerization and activation. That shift matters because ERK activation sits at the center of many growth and survival programs in cancer.
In other words, lactate is not simply a byproduct accumulating in the tumor microenvironment. Here, it behaves like a signaling input that helps push ERK into a more active state, while ERK itself amplifies the very enzymatic activity that modifies it.
A peptide-based way to break the loop
One of the most translational pieces of the study is the development of a cell-penetrating peptide designed to specifically block ERK lactylation. In preclinical testing, this peptide impaired tumor growth in KRAS-mutant cancer models, suggesting the lactylation interface could be a useful therapeutic target in RAS–ERK-driven disease.
That makes the work especially interesting for peptide researchers: it shows how a short, engineered peptide can be used not just to block a receptor or enzyme active site, but to interfere with a disease-promoting modification cycle inside the cell.
Why this matters
Much of the recent cancer-metabolism field has focused on how lactate shapes the immune environment or alters gene regulation. This study expands that picture by showing a direct route from lactate to oncogenic signaling through ERK and GCN5. It also connects metabolic remodeling to a targetable biochemical loop, which may open new paths for combination strategies in tumors that rely on RAS–ERK signaling.
For now, the takeaway is straightforward: lactate may be doing far more than accumulating in tumors. It may be helping to keep a major growth pathway switched on.