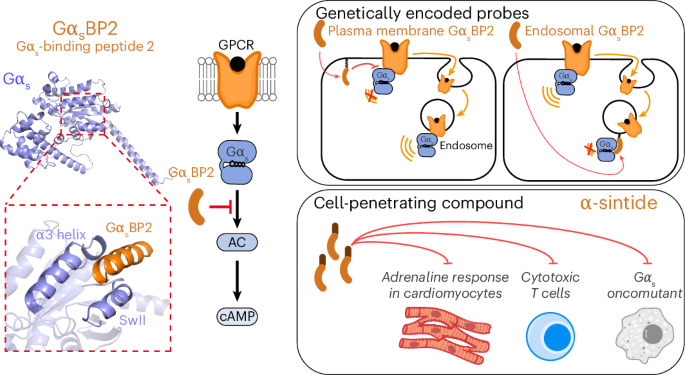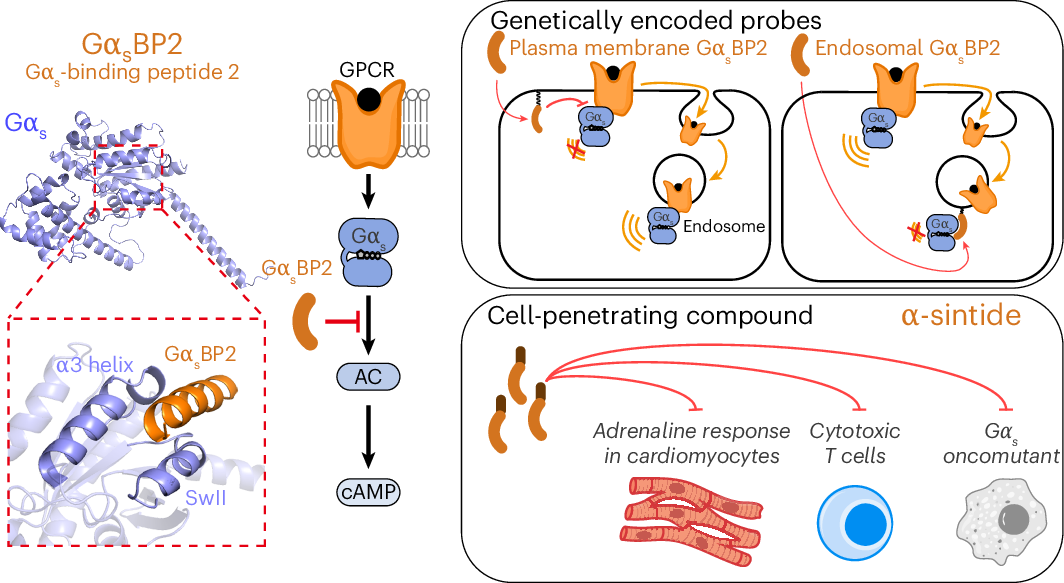
Gαs is one of the best-known switches in GPCR signaling, helping relay signals from cell-surface receptors to downstream effectors. It is also the G protein most often altered in cancer, making it a major focus for both basic biology and therapeutic research.
For years, the dominant model placed Gαs signaling at the plasma membrane. That picture has been steadily challenged by evidence that Gαs can also act from inside the cell, including on intracellular organelles. But proving where and when that signaling occurs has been difficult, largely because existing tools have not offered enough control over location and timing.
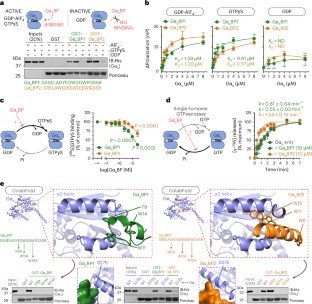
In this study, the authors introduce a new set of inhibitory probes designed to block the effector-binding site of active Gαs in living cells. The toolkit includes genetically encoded probes as well as cell-penetrating compounds, allowing researchers to shut down signaling at specific subcellular sites, at chosen time points, and under a range of experimental conditions.
Using these probes, the team provides direct evidence that Gαs can signal from intracellular organelles. They also report that cancer-associated Gαs variants show distinct signaling behavior in space and time, suggesting that oncogenic mutations may alter more than just signaling strength.
Beyond cancer biology, the work shows that Gαs signaling can be selectively controlled in physiologically relevant settings such as cardiac and immune cells. That raises the possibility of using these tools to dissect normal signaling pathways, disease mechanisms, and potential drug responses with far greater precision than before.
Overall, the study opens a new route for investigating GPCR biology: not just asking whether Gαs is active, but where it is active, when it is active, and how that context shapes its biological effects.