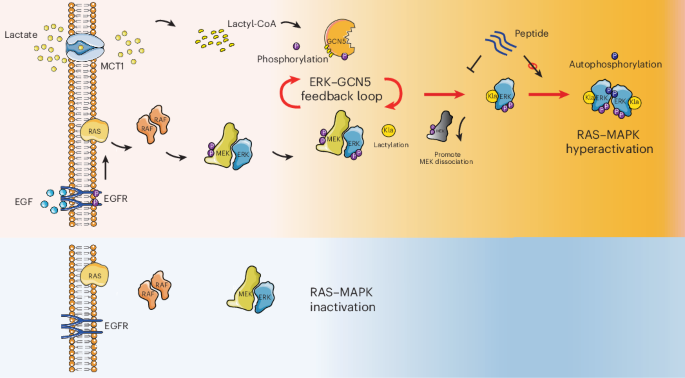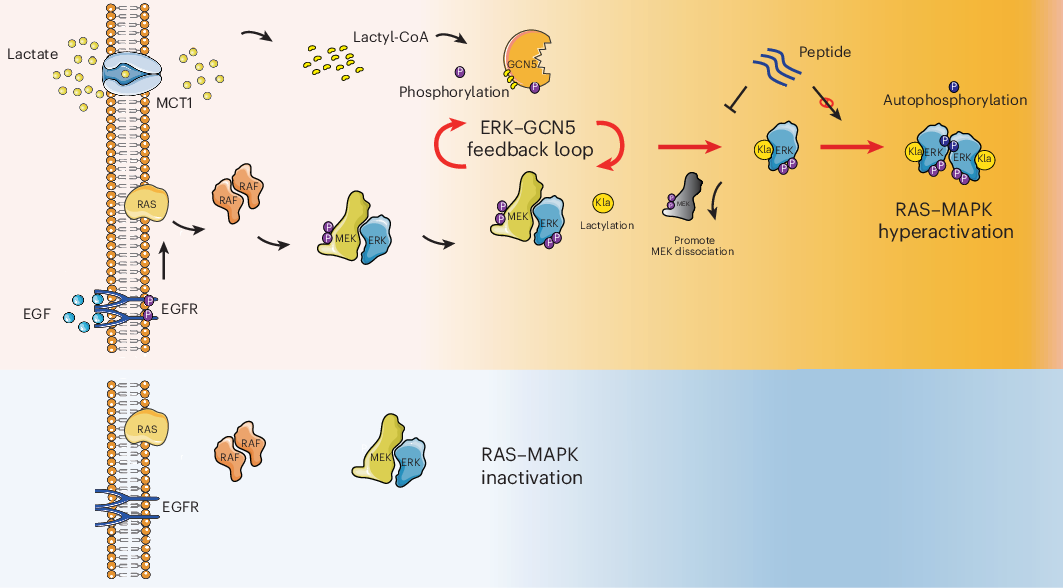
Cancer cells are well known for their high rates of glucose use and lactate production. For years, that excess lactate has been viewed mainly as a metabolic byproduct with broader effects on the tumor microenvironment. A new study adds another layer: lactate may also act directly inside cancer cells by modifying a key growth pathway.
In this work, researchers report that lactate stimulates the MAPK pathway through ERK lactylation. They identify GCN5 as the enzyme that installs this modification on ERK, making it a lactyltransferase in this context. Once ERK is activated, it feeds back on GCN5 by phosphorylating it, which increases GCN5’s ability to lactylate ERK again. The result is a self-reinforcing loop that can amplify lactate-driven cancer progression.
A modification at ERK K231 changes signaling behavior
The study points to a specific site on ERK, lysine 231, as the key lactylation residue. According to the authors, adding a lactyl group at this position weakens ERK’s interaction with MEK. That shift appears to favor ERK dimerization and activation, helping push the pathway into a more active state.
This is notable because ERK sits at the center of a signaling network that controls cell growth, survival, and division. In tumors driven by RAS–ERK signaling, even modest changes in ERK regulation can have outsized effects on proliferation and malignancy.
A peptide strategy to interrupt the loop
Beyond mapping the mechanism, the team developed a cell-penetrating peptide designed to specifically inhibit ERK lactylation. In preclinical testing, the peptide reduced tumor growth in KRAS-mutant cancer models, suggesting that blocking this modification could be a useful way to dampen hyperactive RAS–ERK signaling.
The findings place lactate in a more direct signaling role than many cancer researchers may have expected. Rather than acting only as fuel or an environmental cue, lactate can also shape intracellular kinase activity through protein lactylation and phosphorylation crosstalk.
Why this matters
If these results hold up in broader settings, they could help explain how tumors turn metabolic rewiring into stronger growth signals. They also open the door to a new therapeutic angle: targeting ERK lactylation itself, rather than only upstream oncogenic drivers.
For cancers with active RAS–ERK signaling, that could be especially relevant. The study suggests that interrupting the lactate-GCN5-ERK circuit may offer a way to blunt tumor progression by breaking a feedback loop that cancer cells use to keep the pathway switched on.